Introduction
Ludwig’s angina was first reported by Wilhelm Frederick von Ludwig in 1836. It is a severe and quickly spreading cellulitis characterized by significant involvement of the submandibular, sublingual and occasionally submental space. This condition is potentially fatal if not treated aggressively and promptly due to the cellulitis leading to airway compromise. 1,2 Prior to the introduction of antibiotics in the middle of the 20th century, mortality rates were more than 50%. 3,4
Most cases of Ludwig’s angina documented occur more commonly in adult males (aged 20-60) and occur due to odontogenic infection (70-90%) with mandibular molars (uni or bilateral involvement) as the main cause. 2-11 If and when Ludwig’s angina occurs in the pediatric population, odontogenic cause is only 50%. 11 Some other reported causes include salivary gland infection due to sialadenitis, 12 oral tumor infection 13 and infection following intraoral soft tissues surgery. 14 Furthermore, Ludwig’s angina occurs mostly in patients who are otherwise previously healthy with no history of medical conditions and in children, a precipitating etiology may be difficult to determine. 5,6,9,11,15 The most frequently involved microorganism in Ludwig’s angina is streptococci species along with staphylococci species. Diagnosis of the condition is normally made through history and physical examination but other findings include fever and elevation of the white blood cells. 2 The patient presented here shows a severe case of a Ludwig’s angina found in a healthy adolescent male, an age group in which the condition is less common.
Case Report
A 16-year-old male was airlifted to British Columbia Children’s Hospital (BCCH) pediatric intensive care unit (PICU) overnight from a First Nations community in the northern part of the province. The patient’s medical history was significant for ventricular septal defect repair at age five with residual patch leak present, not on any medications and no known drug allergies.
The patient’s history of present illness was five days of significant tooth pain on the lower right side of his face, which slowly progressed to extensive bilateral neck and facial swelling, difficulty breathing, dysphonia, and fever. The patient initially sought medical attention at the community hospital where a non-contrast computerized axial tomography (CT scan) showed diffuse soft tissue swelling and narrowing of airway. The patient had an oxygen saturation of 92% on room air and was subsequently administered oxygen via nasal canula. The patient was intubated with a 6.5 mm cuffed endotracheal tube and placed on propofol infusion. Intravenous (IV) ceftriaxone, metronidazole and penicillin G were given empirically. He was then airlifted under sedation to the PICU at BCCH for further care (Fig. 1).
Fig. 1
Initial presentation of the patient upon arrival to the Pediatric ICU after being airlifted from the community hospital.

Upon arrival to the PICU, the patient was started on morphine and continued on IV infusion of ceftriaxone, metronidazole and penicillin G. The patient was kept intubated and placed on positive pressure support. Initial vital signs were a heart rate of 105 bpm, blood pressure 126/49 mmHg, and a temperature of 38.3°C. A complete blood count was performed that revealed a white blood cell count of 16 x109/L, up from an initial count of 13.4 x109/L at the community hospital.
Physical examination revealed bilateral submandibular and submental swelling that was tender to palpation, along with sublingual space elevation seen intraorally. Erythema was appreciated in the anterior neck down to the sternal notch, the bilateral submandibular and submental areas (Fig. 2). The patient had poor oral hygiene and carious dentition. A CT scan with contrast was taken which showed multiple loculations in the bilateral submandibular, submental, lateral pharyngeal spaces, and tracking down to the supraclavicular area (Figs. 3a-3d). The pre-operative diagnosis was bilateral submandibular, submental, sublingual, pterygomandibular and pre-tracheal space infection leading to a presentation of Ludwig’s angina.
Fig. 2
Erythema extending from the neck down past the sternal notch

Fig. 3a
Pre-surgical axial CT, showing abscess formation in the bilateral pterygomandibular space.

Fig. 3b
Pre-surgical axial CT, showing abscess formation in the bilateral submandibular space and the submental space.

Fig. 3c
Pre-surgical coronal CT, showing abscess formation in the submental space.

Fig. 3d
Pre-surgical axial CT, showing abscess formation in the pretracheal space.

The patient was brought to the operating room in conjunction with otolaryngology for incision and drainage of bilateral submandibular, submental, sublingual, pterygomandibular, pretracheal and supraclavicular spaces and the extraction of the severely carious left and right second mandibular molars (teeth #3.7 and 4.7) on the same night which he was admitted. Multiple areas of abscess in the oral and neck areas were found and approximately 50 ml of purulence was drained. Four penrose drains were placed (one in the submental area, two submandibular areas and one in the lower neck), and a dressing was placed (Fig. 4). The patient was kept intubated and was transferred back to the intensive care unit in a stable condition.
Fig. 4
During the initial surgery four extraoral penrose drains were placed in the submental, submandibular and tracheal areas.
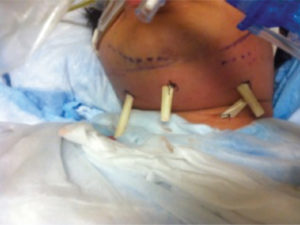
The patient remained intubated and on positive pressure support for two full days following surgery. The neck dressing was changed two to three times daily, the penrose drains were slowly advanced inch-by-inch daily, and were irrigated with saline. The white blood cell count on post-op day one was 15 x109/L (down slightly from 16 x109/L the prior day) and dropped significantly to 10.9×109/L on post-op day two, which suggested the patient was responding well to the treatment. On post-operative day two the cultures from the submental and submandibular abscesses and the extraction sites revealed gram positive cocci (normal oropharyngeal flora, S constellatus, C.albicans, and C. tropicalis) which were determined to be sensitive to clindamycin and penicillin. With the result of the cultures and sensitivities, the Department of Infectious Diseases decided to continue on the current course of antibiotics.
The patient was taken off pressure support on post-operative day three and was extubated. The patient was continued on the same IV antibiotics and a daily chlorhexidine rinse was prescribed. The patient reported feeling much better.
On post-operative day five, three of the four penrose drains had been removed or had fallen out as the neck dressing was changed, with only the submental drain remaining. Iodoform gauze was placed and removed daily in the neck incisions that had no drain present. Although swelling had decreased and the patient reported a significant improvement, the patient had vomited overnight and once again became febrile on post-operative days six and seven. Swelling, especially in the pre-tracheal region remained. The patient’s white blood cell count had elevated to 16.7 x109/L. A repeat CT with contrast was ordered which revealed several radiolucent areas remaining in the lower neck region. The CT showed no obvious loculations in the oral areas, with only loculations apparent in the lower neck and supraclavicular area (Figs. 5a-5d).
Fig. 5a
Post-op axial CT, showing abscess drained from the bilateral pterygomandibular space.

Fig. 5b
Post-op coronal CT, showing resolution of the submental abscess with penrose drain in place.

Fig. 5c
Post-op axial CT, showing the pre-tracheal abscess is still present.

Fig. 5d
Post-op axial CT, showing the infection spreading into the mediastinum.

Subsequently, a second surgery by the otolaryngologist was planned. Prior to the second surgery, it was determined that the six maxillary anterior teeth (#1.3, 1.2, 1.1, 2.1, 2.2, 2.3) were severely carious and required extraction.
On post-operative day eight another surgery was performed with otolaryngology for the extractions, and incision and drainage. Once under general anesthesia, the patient’s anterior teeth were removed. An incision and drainage was completed at the midline thyroid level with copious amounts of purulence tracking from midline submental/submandibular spaces to the level of the thyroid. Pre-thyroid spaces were drained and septae were broken down. Iodoform gauze was placed in the incisions along with penrose drains.
The patient remained in the hospital for a week following the second surgery as the swelling slowly decreased (Fig. 6). In addition, the patient’s white blood cell count fell significantly to 5.4×109/L two days after the second surgery. Penrose drains and iodoform gauze were removed several days after the surgery. IV antibiotics were continued until day 14 in hospital. On day 14, white blood cell count was reported at 4.3 x109/L (a further drop from the 5.4×109/L number the week prior). The patient was discharged from the hospital on day 14 and placed on oral antibiotics (metronidazole and penicillin V) for an additional two weeks. The patient was scheduled for a follow-up visit with the oral surgeon but did not show up for the appointment. It is believed that he moved back home with no issues.
Fig. 6
Post-op Day 12. Edema and erythema significantly decreased following surgical intervention.

Discussion
Ludwig’s angina is described as a severe and quickly progressing cellulitis involving multiple facial spaces of the face and neck. The submandibular spaces bilaterally are the most commonly affected with the possibility of further spread to deeper spaces. Infection of the submandibular space can itself cause airway compromise, as the floor of the mouth and tongue are displaced superior and posterior. Further spread of the infection to the lateral pharyngeal and retropharyngeal spaces may cause more significant respiratory complications. Involvement of the cardiovascular system may also be possible as the infection continues to track downwards to the mediastinum and pericardium. 2,5,9,11 Our case is a typical presentation of a Ludwig’s angina; however, it is in an atypical age group. The possibility of cardiac involvement was considered due to the significant spread of infection in the lower neck but was ruled out by the radiologist.
Signs and symptoms associated with Ludwig’s angina include mild pain/swelling in the upper neck, lower posterior tooth pain, trismus, floor of mouth swelling and superior/posterior displacement of the tongue, tachycardia, fever, and malaise. Increasing airway obstruction also occurs due to continued swelling in the soft tissues of the neck. Dyspnea, dysphagia and drooling may occur depending on severity. Clinically, this obstruction is seen as an extension of the neck in the so-called “sniffing position”. This can be lifethreatening as patients progress to tachypnea, stridor, cyanosis followed by death if the airway is not managed immediately. 2,4,7,11,15 Infection of the surrounding tissues in the area may also be possible causing osteomyelitis and necrotizing fasciitis. 3,16 Although no significant involvement of the surrounding tissue was noted during the treatment course of our patient, airway obstruction was a primary issue, as the soft tissue swelling of the neck appeared to compromise the patient’s airway quite early after presentation of tooth pain.
Diagnosis of the Ludwig’s angina is mainly clinical and is based on criteria by Ludwig and Grodinsky in 1939. They described the condition as a cellulitis of the submandibular space potentially involving more than one neck space causing swelling of the floor of the mouth and posterior/superior displacement of the tongue. The infection does not commonly affect glandular tissues and spreads by continuity instead of through the lymphatic or vascular system. 5,9,10 Further helpful diagnostic tests may include hematologic analysis, and diagnostic imaging such as plain radiographs, ultrasound, CT and MRI (especially for severe cases which have potential to involve multiple systems). While important, these diagnostic tests should not delay definitive treatment. 2,4,6,7,10
Treatment of Ludwig’s angina involves immediate maintenance of the patient’s airway either by endotracheal intubation (if possible) or by tracheostomy or cricothyroidotomy, followed by antibiotic therapy and surgical intervention. Surgical intervention via incision and drainage is paramount as antibiotics alone are not sufficient treatment with such severe infections. Antibiotic therapy includes broad-spectrum antibiotics such as penicillin, clindamycin, cephalosporins, and metronidazole to cover aerobic and anaerobic bacteria, which may be commonly found in the oral cavity. Streptococcus and staphylococcus species are the most commonly found bacteria in cases of Ludwig’s angina. 2 Surgical intervention includes decompression of the affected facial spaces through incision and drainage of purulence if present and is indicated if medical management does not yield significant improvement or localized abscess formation is evident. 2-5,9-11,15
A multi-disciplinary team consisting of specialists from PICU, otolaryngology, infectious disease, radiology, oral surgery, and pediatric dentistry managed our patient. Treatment included endotracheal intubation; IV antibiotics, along with removal of the mandibular second molars and surgical incision and drainage both intraorally and extraorally. The drains extended extraorally from the submental and submandibular area through the submental and submandibular spaces and into the oral cavity. The patient required a second surgery for incision and drainage with penrose drains extending from the submandibular space down to incisions made at the level of the thyroid. This second surgery was required to adequately drain the pre-tracheal region. It was suspected that the initial incision and drainage did not adequately reach all the abscesses. Drains are vital to the treatment of Ludwig’s angina, but drains not placed in the appropriate positions may prevent the complete resolution of the bacterial infection. To ensure adequate drainage of loculations and adequate placement of drains, a post-op CT scan would be beneficial for severe cases of Ludwig’s angina, where significant swelling has progressed to the lower neck region. It is also very important to note that infections are not static and require constant re-evaluation until completely resolved. In aggressive infections, it is not uncommon to have patients require multiple operating room visits to redo the incision and drainage.
Conclusion
The present case describes a typical but severe case of Ludwig’s angina in a relatively atypical age group. This is a life threatening condition, which requires early intervention along with an interdisciplinary team when multiple systems are involved. While relatively uncommon in the pediatric population, dentists should be aware and understand the urgency of immediate treatment when it does present in young individuals. OH
Oral Health welcomes this original article.
References
1. Candamourty R, Venkatachalam S, Babu MR, et al. Ludwig’s Angina–An emergency: A case report with literature review. J Nat Sci Biol Med, 2012; 3(2): 206-8.
2. Spitalnic SJ, Sucov A. Ludwig’s angina: case report and review. J Emerg Med, 1995; 13(4): 499-503.
3. Buckley MF, O’Connor K. Ludwig’s angina in a 76-year-old man. Emerg Med J, 2009; 26(9): 679-80.
4. Wasson J, Hopkins C, Bowdler D. Did Ludwig’s angina kill Ludwig? J Laryngol Otol, 2006;120(5): 363-5.
5. Marcus BJ, Kaplan J, Collins KA. A case of Ludwig angina: a case report and review of the literature. Am J Forensic Med Pathol, 2008;29(3): 255-9.
6. Gaspari RJ. Bedside ultrasound of the soft tissue of the face: a case of early Ludwig’s angina. J Emerg Med, 2006; 31(3): p. 287-91.
7. Hughes D, Holt S, Kman NE. Ludwig’s angina triaged as an allergic reaction. J Emerg Med, 2013; 45(5): p. e175-6.
8. Kassam K, Messiha A, Heliotis M. Ludwig’s Angina: The original angina. Case Rep Surg, 2013: Article ID 974269
9. Britt JC, Josephson GD, Gross CW. Ludwig’s angina in the pediatric population: report of a case and review of the literature. Int J Pediatr Otorhinolaryngol, 2000; 52(1):79-87.
10. Costain N, Marrie TJ. Ludwig’s Angina. Am J Med, 2011;124(2): 115-7.
11. Lin HW, O’Neill A, Cunningham MJ. Ludwig’s angina in the pediatric population. Clin Pediatr (Phila), 2009; 48(6): 583-7.
12. Honrado CP, Lam SM, Karen M. Bilateral submandibular gland infection presenting as Ludwig’s angina: first report of a case. Ear Nose Throat J, 2001; 80(4): 217-8, 222-3.
13. Fischmann GE, Graham BS. Ludwig’s angina resulting from the infection of an oral malignancy. J Oral Maxillofac Surg, 1985; 43(10): 795-6.
14. Lin HW, O’Neill AO, Rahbar R, et al., Ludwig’s angina following frenuloplasty in an adolescent. Int J Pediatr Otorhinolaryngol, 2009; 73(9): 1313-5.
15. Saifeldeen K, Evans R. Ludwig’s angina. Emerg Med J, 2004; 21(2): 242-3.
16. Chueng K, Clinkard D, Enepekides D, et al, An unusual presentation of Ludwig’s angina complicated by cervical necrotizing fasciitis: A case report and review of the literature. Case Rep Otolaryngol, 2012; Article ID 931350.
About the Authors
 Dr. Cheung is a staff pediatric dentist at the BC Children’s Hospital in Vancouver, British Columbia, and is a clinical associate professor, Faculty of Dentistry, University of British Columbia, Vancouver, British Columbia.
Dr. Cheung is a staff pediatric dentist at the BC Children’s Hospital in Vancouver, British Columbia, and is a clinical associate professor, Faculty of Dentistry, University of British Columbia, Vancouver, British Columbia.
 Dr. Choi is a staff oral and maxillofacial surgeon at the BC Children’s Hospital in Vancouver, British Columbia, and is a clinical assistant professor, Faculty of Dentistry, University of British Columbia, Vancouver, British Columbia.
Dr. Choi is a staff oral and maxillofacial surgeon at the BC Children’s Hospital in Vancouver, British Columbia, and is a clinical assistant professor, Faculty of Dentistry, University of British Columbia, Vancouver, British Columbia.
 Dr. Papadopoulos is a former hospital dental resident at the BC Children’s Hospital in Vancouver, British Columbia. He is currently a graduate orthodontic specialty resident at Western University in London, Ontario.
Dr. Papadopoulos is a former hospital dental resident at the BC Children’s Hospital in Vancouver, British Columbia. He is currently a graduate orthodontic specialty resident at Western University in London, Ontario.
 Dr. Wong is a former pediatric dentistry specialty graduate student at the University of British Columbia in Vancouver, British Columbia. He is currently practicing pediatric dentistry in Edmonton, Alberta and is a clinical assistant professor, Department of Dentistry, Faculty of Medicine and Dentistry, University of Alberta, Edmonton, Alberta.
Dr. Wong is a former pediatric dentistry specialty graduate student at the University of British Columbia in Vancouver, British Columbia. He is currently practicing pediatric dentistry in Edmonton, Alberta and is a clinical assistant professor, Department of Dentistry, Faculty of Medicine and Dentistry, University of Alberta, Edmonton, Alberta.












