Introduction
According to the Seventh European Workshop, 15-30% of all implants placed will develop peri-implantitis. Three million people have implants and this number is growing by 500,000 a year. Thus, peri-implantitis cases will grow up to 450,000 with an additional 75,000 which are expected to occur annually. 1 Clinicians will need to find appropriate treatment modalities to solve this exponentially increasing problem.
Treatment modalities have traditionally consisted of treating with chemical means, phosphoric acid, citric acid and tetracycline. These means have produced inconsistent results at best. In 2006, Dr. Robert Miller published in “Implant Dentistry” on the use of Er,Cr:YSGG to disinfect the implant surface with the use of laser for disinfection. The use of laser therapy has shown to have a greater effect than chemical means. According to Miller’s publication, the demonstrated depth of disinfection to over 1000 microns is possible, compared to around 100 microns 2 when chemicals are used.
Treatment by dental laser has not produced consistent results by itself. On April 8th, 2017, the annual meeting of the Academy of Laser Dentistry invited companies to participate in developing a consensus on a treatment of peri-implantitis that can work consistently. Companies that were invited were: Convergent, Biolase, Lightscalpel, Fotona and Millenium. Companies that elected to participate were: Convergent, Biolase, Lightscalpel and Fotona. Various criteria were discussed, and the group gave treatment recommendations on using lasers as an adjunct for peri-implantitis.
A presentation using the 9300nm CO2 (Convergent dental, Boston, Massachusetts) laser featured Edward R Kusek. Dr. Kusek is a Diplomate of ABOI, FAAID MALD MAGD. Dr. Kusek presented the use of Oral DNA as a pre-determinate to find pre-disposition for peri-implantitis/gum disease. Kusek showed two separate cases in which patients presented with advanced periodontal disease (Fig. 1), but their Oral DNA did not indicate a predisposition to periodontal disease.
Kusek reported a correlation between patients with high risk genotypes and higher pre-disposition for some type of peri-implant disease.
Kusek reported on a detailed study he had conducted in his office using anaerobic swabs around infected implants. The study consisted of swabbing three different times. The first swab was taken when the soft tissue was reflected around the granulation tissue. Second swab was taken after all granulation tissue was removed around the failing implants and a titanium brush had been used to clean the entire surface of exposed implant threads. The final swab was completed after using irradiation from the 9300nm CO2 laser. The swabs were then taken to a local lab to plate. This lab was instructed to find periodontal pathogens (Porphyromonas gingivalis, Prevotella intermedia, Beta Hemolytic streptococci, Cam-pylobacter-Wolinella, Capnocytophaga species, Fusobacterium, Peptostreptococcus microns, enteric gram-negative rods, Enterococcus species, Provetella melaninogenica and non-pigmented Prevotella.), if present around a failing implant. A majority of cases showed high levels of bacteria on the first swab, moderate amounts on the second swab and all, but one, showed no bacterial growth on surfaces irradiated by the 9300nm CO2 laser (Fig. 2). The one of the four plated cases showing high amounts of bacterial growth was a heavy smoker (Fig. 3).
Kusek presented eight cases of patients treated with the CO2 laser set to different energy levels. Two cases demonstrated that treatment using lower energy values had treatment failures and recurrence of infection. When higher energy levels around 14 W were used for at least 40 seconds (over 550J of energy), all cases showed success. Subsequently, the two failed cases have been re-treated with higher energy values and are now successful, with one case having a post-op time of over one-year.
Fig. 1

Fig. 2

Fig. 3

Laser-Assisted Peri-implantitis Treatment Protocol
- Flap extention is made two teeth beyond the infected site using a 9300nm CO2 laser with only one releasing incision in a non-aesthetic position (Fig. 4).
- Elevation of the flap to expose any granulation tissue over the implant body.
- A surgical hand-piece is used to remove the granulation tissue around the threads of the implant body (Fig. 5). The treatment of three- or four-wall defects has a good prognosis when at least half of the implant body is fixed into solid alveolar bone.
- The use of a titanium brush (Salvin Dental Specialties, Charlotte, NC) to scrub the surface of the implant to the point where there are no tissue tags on the implant surface with visible loupes (Fig. 6).
- The use of 9300nm CO2 laser (Convergent dental Boston, Massachusetts.) set to 60% police which allows an average of 14 W of energy to the surface of the implant body. The laser tip is positioned approximately 15 mm away from the implant surface with 1 mm
spot size and 100% water mist (Fig. 7). The use of a water cooling system is necessary to keep the implant and bony wall from overheating the site, which would cause surrounding bone to necrotize. 3 - The use of Er,Cr:YSGG laser (Biolase, Irving, CA) around the bony walls with setting of 4.5W 31A/21W with a MZ8 (800 micron) tip to create bleeding points around the bony walls (Fig. 8). This stimulates fibroblasts and aids in the healing process (micro-RAP phenomenon). 4
- Placement of Bio Oss (Geistlich Pharm AG, Wolhusen, Switzerland) bone graft material soaked in PRF (platelet rich fibrin) plasma and cut into small pieces to place around the implant body (Fig. 9) followed by coverage with an extended collagen interlocking membrane placed to cover the interproximal portion of the implant.
- Placement of a PRF membranes (Intra-spin, Intra-lock, Boca Rotan, Florida) to cover the Collagen membrane and implant body (Fig. 10).
- The area is sutured using 3.0 PTFE (Salvin Dental Specialties, Charlotte, NC) sutures with a deep horizontal mattress for coronally re-positioned flaps and followed by interrupted sutures to attain tight primary closure.
- The use of Er,Cr:YSGG laser 4.0W, 20Hz, 31A/0W to de-epithelialize the tissues up to 1 cm past the entire surgical site 5 to aid in its healing (Fig. 11).
- Sutures are removed around two weeks post-op.
- The use of photo-bio-modulation (PBM) first at the time of surgery, again one week after surgery, and a final time after two weeks or at the suture removal appointment.
- The patient needs to keep the area clean by manual toothbrush and should not use oral irrigators. Two months of healing is recommended before an oral irrigator or electric toothbrushes can be used (Sonic Care, Philips Care, Andover, MA/ Oral B Cincinnati, OH). The tissues appearance at six weeks post-op can be seen in Figure 12.
- Radiographs can be taken at a recall appointment after six months. Using this protocol, Kusek has reported 100% success rate for treatment of three and four wall defects.
Fig. 4

Fig. 5

Fig. 6

Fig. 7

Fig. 8

Fig. 9
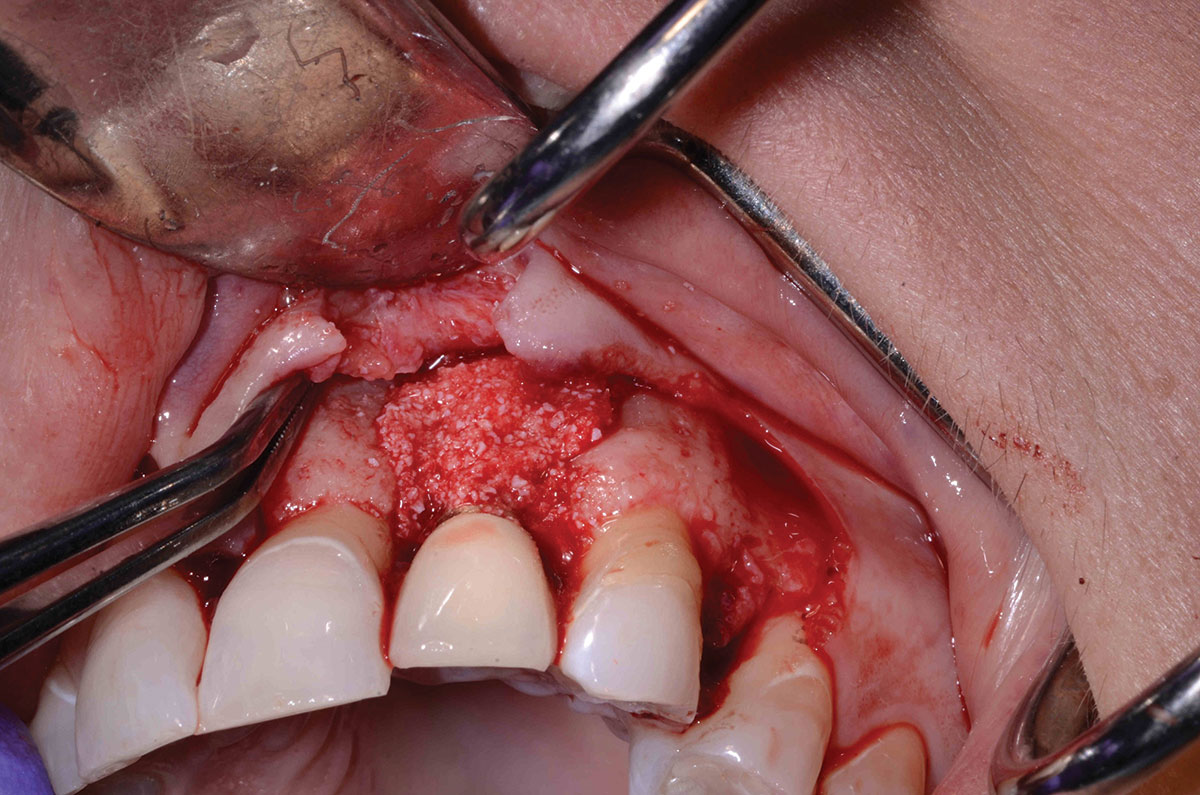
Fig. 10

Fig. 11
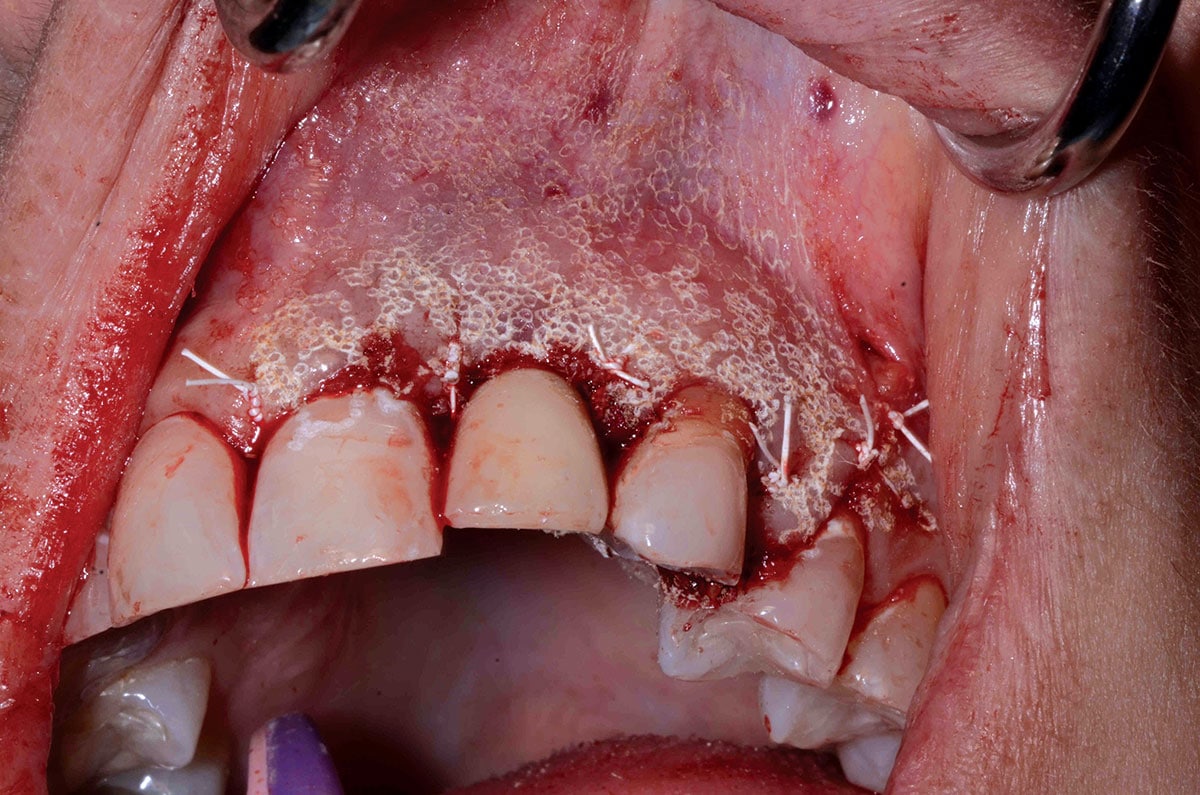
Fig. 12

Summary
- Flap reflection is necessary to gain access when treating a three- or four-wall defect. These are the only defects that have been shown to be treatable.
- Biofilm must be removed by either a Glycerin Air Polisher (Air Flow or HuFriedy) or Titanium brush (Salvin).
- Use of either Erbium or CO2 lasers is recommended. These have proven to be the best lasers to detoxify implant surface without creating excess heat that would lead to de-bonding of the implant/bone contact. The least effective lasers for this treatment are diodes and Nd:YAG, as they create excess heat on the metal implant surface.
- Decortication of the bone surrounding the implant (Erbium) or piezo and de-epithelization of soft tissues outside of the flap (CO2 or Erbium) should be performed.
- Use of Biologics (PRF) in combination with particulate grafting (usually Xenograft) or slowly resorbable graft material is recommended.
- Before closing the site, a Collagen membrane that is interwoven to prevent tissue invagination and then a PRF membrane covering the Collagen membrane, should both be affixed. 6 OH
Oral Health welcomes this original article.
References
- Berglundh T, Claffey N, Lindhe J et.al. Peri-implant diseases; Consensus report of the sixth European workshop on periodontology. J Clin Periodontol 2008;35(8):282-285.
- Miller RJ. Treatment of the Contaminated Implant Surface using the Er,Cr:YSGG Laser. Implant Dentistry. 2004. June 13(2): 165-170.
- Mang T, Rogers S, Keinan D, Honma K, Baier R. Antimicrobial photodynamic therapy (aPDT) induction of biofilm matrix architectural and bioadhesive modifications. Photodiagnosis Photodyn Ther. 2016 Mar;13:22-8.
- Verna C, Regional Acceleratory Phenomenon. Front Oral Biol. 2016:18:28-35.
- Kusek ER. Soft tissue management following implant placement. Perio/Implant. Product showcase A-B.
- Kusek et al. Peri-implant Disease Management. Dentistry Today. June 2018 to be published.
About the Author
 Dr. Edward R. Kusek is a Diplomate of American Board of Oral Implantology/Implant Dentistry, Fellow of American Academy of Implant Dentistry, Masterships in Academy of General Dentistry and Academy of Laser Dentistry, Recognized Course provider from Academy of Laser Dentistry since 2012. Diplomate of International Congress of Oral Implantology.
Dr. Edward R. Kusek is a Diplomate of American Board of Oral Implantology/Implant Dentistry, Fellow of American Academy of Implant Dentistry, Masterships in Academy of General Dentistry and Academy of Laser Dentistry, Recognized Course provider from Academy of Laser Dentistry since 2012. Diplomate of International Congress of Oral Implantology.
RELATED ARTICLE: An Underutilized Treatment Modality in the Dental Industry Low Level Laser Therapy
Follow the Oral Health Group on Facebook, Instagram, Twitter and LinkedIn for the latest updates on news, clinical articles, practice management and more!












