Two studies were undertaken to evaluate the retention of tooth whiteness, both objective and subjective, and the perceived smoothness of the treated tooth surfaces with a single application of an innovative product that incorporates both a tooth whitening gel and a whitening sealant. Another objective was to determine the perceived (subjective) retention of whiteness retention and smoothness over the course of the 24 hours following application as compared to matching placebos.
The first study was a two-day, parallel, blind, two cell clinical trial of 2-Step Supersmile Whitening Bolt (Fig. 1). The results of this trial are reported in Part A. Subsequently, 1-Step Supersmile Whitening Bolt (Fig. 2), an analogous, but more convenient product was developed by the manufacturer, Robell Research Inc. (New York, NY). The scheduled introduction of the more user-friendly 1-Step Supersmile Whitening Bolt design coincided with the COVID 19 pandemic; therefore, it was not possible to perform a similar clinical trial for the product. Instead, a laboratory evaluation (Precedent Research Inc., Fort Wayne IN) of extrinsic stain inhibition compared the 2-Step Supersmile Whitening Bolt formula and the 1-Step Supersmile Whitening Bolt formula to an untreated negative control. The results of this evaluation are reported in Part B.
Fig. 1

Fig. 2

PART A
Objective
The objective of this two-day parallel, blind, two cell clinical trial was to determine the retention of tooth whiteness, both objective and subjective, and the perceived smoothness of the treated tooth surfaces with a single application of 2-Step Supersmile Whitening Bolt as compared to matching placebos. Another objective was to determine the perceived (subjective) retention of whiteness retention and smoothness over the course of the 24 hours following product application as compared to matching placebos.
Subjects
Thirty healthy subjects (15 male and 15 female) were entered into the study and all thirty subjects completed the study. There was no exclusion on the basis of baseline tooth coloration. Fifteen individuals from each group were used in the analysis. The average age for the placebo group was 53.8 (SD = 18.7), and the treatment group was 57.7 (SD = 13.9). Females comprised 40% of the placebo group and 60% of the treatment group.
Whitening and Sealing product assignment
Individuals were assigned randomly to the test and control groups, alternating with the order of entry into study. Individuals who were completing the clinical study concurrently were assigned to the same group.
Objective measurement of tooth color
Teeth color was measured both by objective and subjective measurements. For objective measurements, the tooth color of the eight (8) anterior maxillary teeth was measured in a standardized manner. Variability was eliminated with the Vita EasyShade Compact (Vident, Brea, California) intraoral dental spectrophotometer shade-matching device.1,2,3,4 It consists of a light source; a wand to direct the source light to an object and in turn to receive the reflected light from the object; and a spectrometer to determine the received light intensity as a function of wavelength. The EasyShade Compact’s CPU analyzes the spectrophotometer data, determining a shade match to Vita Classic or 3D systems, and displays the results on a touch screen.
For each subject, the shades of the 8 maxillary anterior teeth were each measured separately, in the middle third, at the mesio-distal center of the tooth. These measurements were taken at the Baseline, Post Application, and the Post Staining stages. The shade of each tooth was scored by reading the EasyShade Compact screen and recording the Vita Classic shade reference code. The Vita Classic shade reference code was assigned a score as indicated in the table below. (Fig. 3) The overall change in shade per patient was then obtained by averaging the scores as previously described1,2. Analysis of means (t-test) was employed to compare the mean Vita EasyShade Compact scores at Baseline, Post Application and Post Staining.
Fig. 3

Subjective measurement of tooth color and smoothness
For subjective measurements, the subjects were provided with a printed 10 gradation shade guide (10 the darkest, and 1 the lightest, and 10 the roughest and 1 the smoothest). (Fig. 4) The subjects were then allowed to rank the self-perceived color and the self-perceived smoothness of their teeth on a variable analog scale (VAS) varying from dark to light with the mid-point (5.5) being average.
Fig. 4

Clinical procedure
- After obtaining the Baseline objective and subjective tooth color shades,
- The subjective Baseline smoothness was determined.
- The subjects were then provided with the 2-Step Supersmile Whitening Bolt or the Placebo to use according to the manufacturer’s instructions. These instructions included the supervised application of the test and placebo materials, in sequence, to the 8 maxillary anterior teeth.
- Then, the Post Application objective and subjective tooth color shades were obtained,
- And the subjective Post Application smoothness was determined.
- The subject was asked to consume chromogenic (tooth-staining) foods and chromogenic (tooth-staining) beverages for a period of up to 15 minutes, beginning no less than 3 minutes after Step 5.
- Then, the Post Staining objective and subjective tooth color shades were obtained,
- And the subjective Post Staining smoothness was determined.
- Within 24 hours, the investigators asked about the retention of tooth whiteness after the application of Step 1 and Step 2.
- Within 24 hours, the investigators asked about the retention of tooth smoothness after the application of Step 1 and Step 2.
Objective results Fig. 5
The results of this study show that the single application of the 2-Step Supersmile Whitening Bolt resulted in a very significant improvement of teeth color over the Control during the Application Step – a significant average of 2.2 shades as compared to the Placebo whitening the teeth 0.0 shades.
Fig. 5

Furthermore, the single application of 2-Step Supersmile Whitening Bolt resulted in a very significant maintenance of teeth color over the Control during the Staining Step – the 2-Step Whitening Bolt exhibited a further whitening of 0.15 shades (negligible) as compared to the teeth in the Placebo group darkening a significant average of 2.6 shades with the chromogenic diet.
Subjective results
Subjects were asked to self-score their tooth coloration according to a 10 step VAS printed guide. While quite variable in results, these methods are extensively used in oral health clinical trials to gauge subjects’ self-perceptions. Fig. 6.
Fig. 6

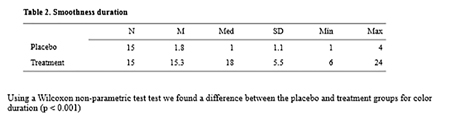
Subjects were asked to self-score their tooth smoothness according a 10 step VAS printed guide. While quite variable in results, these methods are extensively used in oral health clinical trials to gauge subjects’ self-perceptions. Fig. 7
Fig. 7

Within 24 hours, the investigators asked about the retention of tooth whiteness after the application of Step 1 and Step 2. Fig. 8
Fig. 8
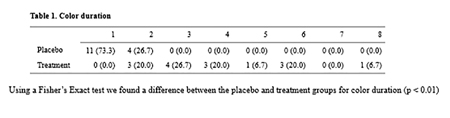
Within 24 hours, the investigators asked about the retention of tooth smoothness after the application of Step 1 and Step 2. Fig. 9
Fig. 9
Discussion
Subjects’ self-perceived color improvement did not always correspond with the magnitude of the objective color change. They tended to underestimate the baseline coloration, to interpret the application coloration changes well, and to recognize the staining effect.
The results of this two-day parallel, blind, two cell clinical trial was to determine the retention of tooth whiteness, both objective and subjective, and the perceived smoothness of the treated tooth surfaces with a single application of 2-Step Supersmile Whitening Bolt as compared to matching placebos. Another objective was to determine the perceived (subjective) retention of whiteness retention and smoothness over the course of the 24 hours following application as compared to matching placebos.
The results of this clinical trial show that the retention of tooth whiteness, both objective and subjective, and the perceived smoothness of the treated tooth surfaces with a single application of 2-Step Supersmile Whitening Bolt as compared to matching placebos were significant, as demonstrated by the data.
Conclusions Part A
- 100% of 2-Step Supersmile Whitening Bolt subjects improved their tooth whiteness upon application.
- After one application, the 2-Step Supersmile Whitening Bolt showed an average of 2.2 shades of whitening as assessed by an objective standardized intraoral dental spectrophotometer shade-matching device.
- After staining, the 2-Step Supersmile Whitening Bolt showed an average of 0.1 shades of whitening, while the Control group exhibited 2.6 shades of staining, as assessed by an objective standardized intraoral dental spectrophotometer shade-matching device.
- After using the 2-Step Supersmile Whitening Bolt once, 80% of participants improved three or more shades as assessed by an objective standardized intraoral dental spectrophotometer shade-matching device.
- In an objective assessment by an objective standardized intraoral dental spectrophotometer shade-matching device 100% of 2-Step Supersmile Whitening Bolt subjects improved their tooth whiteness after one application.
- In an assessment of a subjective survey after using the Supersmile Whitening Bolt once, 80% of participants improved three or more shades.
- Supersmile Whitening Bolt subjects perceived significant improvement in their tooth whiteness for up to 24 hours.
- Supersmile Whitening Bolt subjects perceived significant improvement in their tooth smoothness for up to 24 hours.
PART B5
Purpose
The purpose of this investigation was to evaluate and compare Supersmile’s 1 step whitening and sealing formula (1-Step Supersmile Whitening Bolt) against a separate 2-step whitening and sealing regimen (2-Step Supersmile Whitening Bolt). Each was also compared against an untreated control group in its ability to inhibit extrinsic dental stain while cycling through a 6-hour food stain-producing regimen.
Procedure
The laboratory staining method is a modification of that described by Stookey et al.6 The experimental procedure is described in detail as follows:
Tooth Specimens
Squares of dental enamel from bovine crowns were embedded in self-curing acrylic7. The finished specimens were examined under a dissecting microscope and were discarded if imperfections in the surface enamel were found. The specimens were etched for 60 seconds in 0.2M HCl, followed by a 30-second immersion in a saturated solution of sodium carbonate. After a final etch with 1% phytic acid (60 seconds), the specimens were rinsed and attached to the staining apparatus.
Staining Method
Staining Apparatus
The staining apparatus provided alternating immersion into the staining broth (1.02 g of instant coffee, 1.02 g of instant tea, 50 mls of red wine and 0.75 g of gastric mucin in 200 ml of sterilized trypticase soy broth at 37°C with 50 ml of a 24-hour Micrococcus luteus stain-forming bacterial culture) and air-drying.
Stain Measurement
The intensity of the extrinsic stained pellicle on the teeth was measured by taking diffuse reflectance absorbance readings with a spectrophotometer equipped with diffuse illumination, an 8° viewing angle, and 3-mm aperture. Absorbance measurements were obtained using the tristimulus L*a*b* color space established by the CIE.
Test Products
The products/ingredients were provided by the sponsor.
Study 1
Group 1 – Negative control (no active application)
Group 2 – Test product: 1-Step Supersmile Whitening Bolt SN03742020 mfg 5/27/20
Study 2
Group 3 – Negative control (no application)
Group 4 – Test product: 2-Step Supersmile Whitening Bolt, 2 application (Whitening gel–mfg 6/10/20) (Sealant – mfg 6/10/20)
Treatment
For each study, the specimens were stratified into 2 equal groups of 10 specimens each, with each group having equivalent average inherent (no stain) baseline L*a*b* stain scores. The single day procedure included spectrophotometer readings taken after 2, 4, and 6 hours.
Study 1, prior to immersion into the broth for the beginning of 6-hour exposure, the test enamel slabs were painted with a thin layer of test product (1-Step Supersmile Whitening Bolt) and allowed to air dry for 20 minutes to allow for sufficient adherence.
Study 2, prior to immersion into the broth, the test product (2-Step Supersmile Whitening Bolt) was applied (whitening gel was applied 1st and the sealant 2nd, directly on top of gel) and together were allowed to dry for 20 minutes.
The control specimens were kept in a humidor.
Study 1 and study 2 were run sequentially with each other, thus having separate negative controls.
Interpretation of Data
The difference between the baseline (pre-test) and post-test readings for each color factor (L*, a*, and b*) represented the ability of the test products to reduce existing extrinsic stain and thereby increase tooth whiteness. The individual components of the L*a*b* scale indicated the specific changes in the whiteness (L*), red-green color range (a*), and yellow-blue color range (b*). Data were tabulated using a standard spreadsheet program.
Statistical Evaluation
Efficacy Variables
The primary efficacy variable was extrinsic stain removed for each color factor. The primary endpoint was after 6 hours.
Efficacy Comparisons
The test treatment groups were compared with the negative control to assess relative efficacy.
Statistical Methodology
Data were analyzed by means of a conventional statistics program. For each of the efficacy variables, between treatment comparisons at each time point were made using an ANOVA. All comparisons were performed using a 2-tail test at the 5% level of significance.
Discussion
Study 1: Test product (1-Step Supersmile Whitening Bolt) showed an inhibitory significance after just 2 hours when looking at Δb and ΔE against the control. This significance continued and included ΔL after 4 hours. After 6 hours, ΔL continued to show statistical significance while Δb* and ΔE did not break out (although numerically it showed a noticeable instrumental difference). The test product inhibited stains for several hours.
Study 2: Test product (2-Step Supersmile Whitening Bolt) showed a slight inhibitory effect after only 2 hours when looking at ΔL. After 4 hours, all 3 color components showed statistical significance ΔL, Δb and ΔE against the control. After 6 hours the same trend continued. The test product inhibited stains for several hours.
Conclusions Part B
Both independent in vitro evaluations showed the test products (1-Step Supersmile Whitening Bolt and 2-Step Supersmile Whitening Bolt) exhibited similar inhibitory effects on food stain formation and inhibited stain for several hours. A reasonable expectation from this researcher would be similar in vivo effect.
Overall conclusions
In the clinical study:
- 100% of 2-Step Supersmile Whitening Bolt subjects improved their tooth whiteness upon application with an average of 2.2 shades of whitening as assessed by an objective standardized intraoral dental spectrophotometer shade-matching device.
- After staining, the 2-Step Supersmile Whitening Bolt showed an objective average of 0.1 shades of whitening, while the Control group exhibited a much more significant 2.6 shades of staining.
- After using the 2-Step Supersmile Whitening Bolt once, 80% of participants improved three or more shades as assessed by an objective standardized intraoral dental spectrophotometer shade-matching device.
- In an objective assessment by an objective standardized intraoral dental spectrophotometer shade-matching device 100% of 2-Step Supersmile Whitening Bolt subjects improved their tooth whiteness after one application.
- In an assessment of a subjective survey after using the Supersmile Whitening Bolt once, 80% of participants improved three or more shades.
- Supersmile Whitening Bolt subjects perceived significant improvement in their tooth whiteness for up to 24 hours.
- Supersmile Whitening Bolt subjects perceived significant improvement in their tooth smoothness for up to 24 hours.
In the laboratory study:
- The two independent in vitro evaluations demonstrated that the test products, 1-Step Supersmile Whitening Bolt and 2-Step Supersmile Whitening Bolt exhibited similar inhibitory effects on food stain
formation. - The two independent in vitro evaluations demonstrated that the test products, 1-Step Supersmile Whitening Bolt and 2-Step Supersmile Whitening Bolt inhibited stain formation for several hours.
- It is a reasonable expectation from this research that the in vivo effect would be similar.
The clinical trials have clearly validated the beneficial effects of 2-Step Supersmile Whitening Bolt after one application, both objectively and subjectively. Teeth whitening and staining protection have been substantiated by an objective standardized intraoral dental spectrophotometer shade-matching device. Furthermore, test subjects clearly recognized significant I tooth whiteness improvement after one application of 2-Step Supersmile Whitening Bolt and continued to perceive significant improvements in teeth whiteness and smoothness at 24 hours. The laboratory trial demonstrated the similar actions and effects of 2-Step Supersmile Whitening Bolt and 1-Step Supersmile Whitening Bolt for teeth whitening and inhibition of stain formation. The 1-Step Supersmile Whitening Bolt is more convenient and user-friendly. These research studies therefore make apparent that the 1-Step Supersmile Whitening Bolt will function similarly to the 2-Step Supersmile Whitening Bolt in vivo and will have similar effects and benefits.
Oral Health welcomes this original article.
About the Authors
 Dr. George Freedman is a founder and past-president, American Academy of Cosmetic Dentistry, co-founder, Canadian Academy for Esthetic Dentistry and the International Academy for Dental Facial Esthetics, and a Diplomate and Chair of the American Board of Aesthetic Dentistry. He is Adjunct Professor of Dental Medicine, Western University, Pomona, California and Professor and Program Director, BPP University, London, UK, MClinDent Programme in Restorative and Cosmetic Dentistry. His most recent textbook is “Contemporary Esthetic Dentistry (Elsevier). Author or co-author of 14 textbooks and more than 900 dental articles, Dr. Freedman maintains a private practice limited to Esthetic Dentistry in Richmond Hill, Ontario.
Dr. George Freedman is a founder and past-president, American Academy of Cosmetic Dentistry, co-founder, Canadian Academy for Esthetic Dentistry and the International Academy for Dental Facial Esthetics, and a Diplomate and Chair of the American Board of Aesthetic Dentistry. He is Adjunct Professor of Dental Medicine, Western University, Pomona, California and Professor and Program Director, BPP University, London, UK, MClinDent Programme in Restorative and Cosmetic Dentistry. His most recent textbook is “Contemporary Esthetic Dentistry (Elsevier). Author or co-author of 14 textbooks and more than 900 dental articles, Dr. Freedman maintains a private practice limited to Esthetic Dentistry in Richmond Hill, Ontario.
 Dr Fay Goldstep has lectured extensively on Proactive/Minimal Intervention Dentistry, Soft-Tissue Lasers and Bioactive Dental Materials. She has been a contributing author to four textbooks and has published more than 100 articles. Dr Goldstep serves on the editorial boards of Oral Health Journal, Dental Tribune US Edition, Dental Asia and REALITY. She has been listed as one of the leaders in continuing education by Dentistry Today since 2002. Dr. Goldstep is a consultant to a number of dental companies and maintains a private practice in Richmond Hill, Ontario. She can be reached at goldstep@epdot.com.
Dr Fay Goldstep has lectured extensively on Proactive/Minimal Intervention Dentistry, Soft-Tissue Lasers and Bioactive Dental Materials. She has been a contributing author to four textbooks and has published more than 100 articles. Dr Goldstep serves on the editorial boards of Oral Health Journal, Dental Tribune US Edition, Dental Asia and REALITY. She has been listed as one of the leaders in continuing education by Dentistry Today since 2002. Dr. Goldstep is a consultant to a number of dental companies and maintains a private practice in Richmond Hill, Ontario. She can be reached at goldstep@epdot.com.
 Professor Edward Lynch was elected the most influential person in UK Dentistry in 2010 by his peers. Professor of Restorative Dentistry and Gerodontology of the Queen’s University Belfast as well as Consultant in Restorative Dentistry to the Royal Hospitals, he has also authored more than 500 publications, including chapters in books and referred abstracts. He is a specialist in 3 disciplines, Endodontics, Prosthodontics and Restorative Dentistry. A “wet gloved” academic, Prof. Lynch is a Consultant to the ADA, a spokesperson for the BDA, and the Chairman of the European Experts group on Tooth Whitening. his research team has received10 first prize honours at the IADR.
Professor Edward Lynch was elected the most influential person in UK Dentistry in 2010 by his peers. Professor of Restorative Dentistry and Gerodontology of the Queen’s University Belfast as well as Consultant in Restorative Dentistry to the Royal Hospitals, he has also authored more than 500 publications, including chapters in books and referred abstracts. He is a specialist in 3 disciplines, Endodontics, Prosthodontics and Restorative Dentistry. A “wet gloved” academic, Prof. Lynch is a Consultant to the ADA, a spokesperson for the BDA, and the Chairman of the European Experts group on Tooth Whitening. his research team has received10 first prize honours at the IADR.
 Dr. Patrick C. Hardigan is Associate Dean of Academic Affairs for the Health Professions Division and Executive Dean of Research in the Dr. Kiran C. Patel College of Allopathic Medicine. Dr. Hardigan is the statistician and research advisor for International Health Initiatives and founded NSU’s Statistical Consulting Center. Nationally, he has served as President for the Association for Behavioral Sciences and Medical Education. He is a member of the American Statistical Association and the American Educational Research Association, and the statistical advisor for The Internet Journal of Allied Health Sciences and Practice and the American Journal of Dentistry.
Dr. Patrick C. Hardigan is Associate Dean of Academic Affairs for the Health Professions Division and Executive Dean of Research in the Dr. Kiran C. Patel College of Allopathic Medicine. Dr. Hardigan is the statistician and research advisor for International Health Initiatives and founded NSU’s Statistical Consulting Center. Nationally, he has served as President for the Association for Behavioral Sciences and Medical Education. He is a member of the American Statistical Association and the American Educational Research Association, and the statistical advisor for The Internet Journal of Allied Health Sciences and Practice and the American Journal of Dentistry.
To see more articles from the July/August 2020 issue, please click here!












