Implant treatment has greatly improved the level of care we can offer our patients. According to Misch 2015, many implant retained prosthetics are being installed by a means of intra-oral cementation. 1 The other main fixed prosthesis installation process is by “screwing the already assembled abutment-prosthesis complex”, directly onto dental implants in the mouth. 2
According to a review by Sherif et al., 2014 3 the longevity and failure rates resulting from the two above prosthesis insertion techniques are not significantly different. Considering that implant treatment is becoming ever-more popular and 2.5 million dental implants will be placed in the USA in 2015 (according to an iDataresearch.com report). Even a five percent failure rate would mean 125,000 of these implants will fail over five years. That is a lot of failed dental implants and many of these failed implants will also result in failed implant prosthetics. Failure is very expensive for the clinicians and patients.
There are many identified factors that contribute to implant failure. Failure often presents itself as peri-implant inflammation leading to the loss of peri-implant hard and soft tissue support. Known periodontal pathogens have been isolated from peri-implant tissues with peri-implant disease. 4 Particles of titanium and cement have also been shown to be present in diseased tissues in the peri-implant environment. 5
Zipprich, in his two YouTube videos, has shown that poor fitting implant-abutment connections, which are often positioned at the level of alveolar bone and deep to the gingival tissues, can allow bacteria into the huge internal caverns of the implant. Here they can incubate and invade peri-implant tissues. “Poor fitting” is a relative term that can refer to the fit of any two implant components that allows bacteria with one micron or less diameter to pass between them. 6,7 This can lead to peri-implant disease. 5-9
Wilson (2009) has shown that residual subgingival cement is also a known risk factor contributing to peri-implant disease.10 Indeed the perceived difficulty in controlling the flow and cleanup of subgingival cement has led to the development of a number of different intra-oral cementation techniques to reduce the volume or pressure of excess cement extruding from the margins of the prosthesis during the process of intra-oral cementation. 11-14
Unfortunately underfilling the cement space between the prosthesis and retainer may also pose some problems. Leaving cement voids under the prosthesis can leave space for peri-implant pathogens to incubate and potentially cause peri-implant disease. Also reducing excess cement volume may make it more difficult to locate and clean away excess cement.
Wilson reports that 87 percent of the implants he identified as having peri-implant disease had residual subgingival cement and 71 percent of these cases resolved after removal of this cement. He also mentions that all types of excess cement observed were associated with peri-implant disease. Peri-implant disease was not cement specific. 10
To prevent the negative effects of residual excess cement, some clinicians have abandoned intra-oral cementation techniques, in favor of extra-oral cementation and “assembled abutment-prosthesis installation” by the “screw-in technique. These clinicians appear to be willing to take their chances with ill-fitting implant-abutment connections rather than deal with problems related to residual excess cement. 15
From the above literature and the work of many others, I would suggest that once infected, both the subgingival excess cement and the poor implant-abutment connections can contribute to pathogen mediated peri-implant disease and early failure of the dental implant and its attached prosthesis.
Can we both optimize the implant-abutment connection and prevent the occurrence of residual subgingival cement?
This is possible, by achieving a better understanding of the intra-oral cementation process and implementing the appropriate changes to our techniques. I believe that this new process could significantly reduce the incidence of peri-implant disease and increase the long-term success of the implant-retained prosthesis. I believe this would be especially important to patients that have a history of periodontitis or are otherwise susceptible to assault by periodontal pathogens. I will explain how this can be done below.
After review of the literature and the writing of Misch, 1 the implant industry appears to have difficulty to mass produce implant-abutment connections that are precise enough and strong enough to exclude one micron bacteria from entering the implant body. This problem can worsen if parts are mixed and matched from different manufacturers. 16
In addition, even if we were able to make great implant-abutment connections, we do not have the ability to make our prostheses accurate enough in size, to allow us to install them as “assembled abutment-prosthesis units” directly onto integrated implants, without putting strain onto the implant-abutment connection. 17 Even with optical scans and precision milling, the multiple-unit prosthesis still puts a strain onto the implant-abutment connection that can prevent optimal seating of the abutments. In addition, tight contacts with adjacent teeth, non-parallel implants with internal or external connections and path of insertion issues imposed by adjacent teeth, also add variables that contribute to the misfit of the implant-abutment connection. 17
If you are screwing your “assembled abutment-prosthesis unit” into the intra-oral environment, you are probably contributing to peri-implant disease by increasing the misfit of the implant-abutment connection. 15-17 This allows known pathogens in the deep subgingival space to enter the implant-abutment connection, incubate inside the vast caverns of the implant, and from there invade peri-implant tissues and cause peri-implant disease. 6,7,8
Optimizing the Implant-Abutment connection. To optimize the implant abutment connections we must be able to place the abutments into the intra-oral environment under optimized conditions. Optimized abutment installation conditions include 1) no attached prosthesis (that is either a little too big or too small) to push or pull the abutment off its implant base, 2) no adjacent tooth contacts to push the abutment off its base and 3) good visibility and control of the individual implant sites and 4) ability to check individual abutment seating by X-ray imaging (to avoid gross installation misfits).
This discussion brings us to intra-oral cementation. It is the cement space that allows the clinician to achieve a truly passive connection between the abutment and the prosthesis, when it is installed into the mouth. It is the cement that lutes the two components together and reduces or eliminates strain on the already optimized and tightened implant-abutment connection(s). The problem is that the cementation process can result in residual subgingival cement. 1,14
Understanding the intra-oral cementation process. The cementation process involves a dental cement, a prosthesis and an abutment or an implant-abutment complex. The abutment has a vertical retaining element and a margin on which the prosthesis will rest when it is cemented into place. Some implant-abutment complexes are designed to have the prosthesis rest directly onto the implant margin.
There is usually some cement space between the prosthesis and the abutment. This space is created during the production of the prosthesis by means of a die spacer or specified by means of digital controls for a prosthesis made by a CAD/CAM milling process. This die spacer can be up to 120 microns or more in width. It prevents the prosthesis from binding during cementation process and also allows for the exit of excess cement as the prosthesis is pushed into place. Smaller cement spaces can offer more resistance to the flow of excess cement as it exits from the prosthesis during the cementation process. Smaller cementation spaces were common for cements that had low compressive strength, because they depended on contact or close proximity to their retaining elements to prevent them from crumbling under the forces of mastication. Many of the older cements also displayed a poor ability to prevent cement washout at their margins. Acrylic based cements have much better compressive strength and ability to resist dissolution at their margins.
The margins of the prosthesis and abutment or implant are usually designed to touch when the prosthesis is pushed into place. The touching parts of the margins are designed to minimize the thickness of the cement that fills the space between them and to reduce the size of the cement exposure to the adjacent tissues. The cement is intended to exclude the ingress and proliferation of potential microbial pathogens. It may be of some concern when cement application methods, that do not place cement into the most superior position of the intaglio of the prosthesis, results in trapping air during the cementation process. As air escapes or is forced into the cement it may leave voids at the margin that may harbor pathogens. 18
The process of cementation is a complex hydraulic event by which excess cement is propelled out from between the margins of the retainer and prosthesis. 11-17 The process of intra-oral cementation has many benefits 1,13 but can also result in the injection of excess cement into the peri-implant tissues where it is difficult to locate and clean away. Residual excess cement is a known risk factor for peri-implant disease. 10,14
Many studies have been done in order to better understand the process of intra-oral cementation and its consequences. 12-14 Linkevicius has determined that the deeper the subgingival margin, the bigger the volume of residual excess cement. 19,20 Recently, Dr. Svoboda has developed an “in vitro” models to study the effects of margin design, implant-abutment-prosthesis complex design and gingiva on the flow of excess cement. The results of these studies are presented below. 22-25, 29-31
Many factors contribute to the problem of residual excess cement in the peri-implant environment; these include A) the margin design, B) the position of the margins relative to adjacent tissues, C) the design of the implant-abutment-prosthesis complex and D) the pressure applied by the clinician during the intra-oral cementation process. 17
A) Margin Design
The abutment or implant and complimentary prosthesis margin designs for dental implants have evolved from those used for cemented dental prosthetics on natural teeth. The most common margin designs include the knife or feather edge, tapered, chamfer and shoulder or butt. The feather, tapered and chamfer margins direct excess cement down into the peri-implant tissues. Due to the designs of the standard abutments with their narrow profiles, Dr. C.E. Misch favoured the feather margin. 21 This is probably one of the worst designs for directing cement deep into the tissues during the intra-oral cementation process. The butt margin can also project cement into the tissues, depending on its angle and location of the gingiva. 22
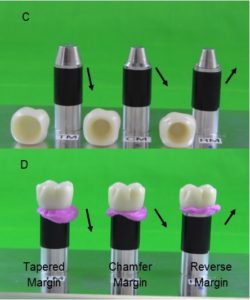
Dr. Emil Svoboda, has designed the “Reverse Margin™” that redirects the cement up and out of the tissue spaces rather than into them. 22-25 Figures 1 A & B show the effect of the inflected margin design on the destination of excess cement. The Reverse MarginTM has propelled the cement into an upwards direction and is mainly above the dotted line while the other margin designs (Tapered and Chamfer) propelled the cement in a downwards direction. Watch the “Reverse Margin™, Margin Design is Important” Video, 25 to observe the cement flowing out from between the crown margins during the cementation process. Figures 1 C & D are derived from this later video using solid zirconium crowns. Note that the cement expressed upwards by the Reverse Margin™ design of the crown and abutment stays above the black tape marker while the other margins direct the cement downwards. This effect of the margin design on the direction of expressed excess cement has been repeated many times and the results do not vary. RelyX Ultimate is less viscous than DAP and still follows the same pattern. Using the Reverse Margin™ design is the first step in redirecting cement out of the tissues rather than into them, thus preventing residual subgingival cement.
Fig. 1 A&B
Figure 1 A—Shows 3 aluminum rods with different margin designs. B—Shows the rods with clear plastic crowns pushed into place. The arrows indicate the direction of the margins and the cement flow. The top dotted line is in line with the tops of the crowns while the bottom line is in line with the margins.

Fig. 1 C&D
Figure 1 C—Shows 3 aluminum rods with different margin designs. D—Shows the rods with zirconia crowns cemented into place. The arrows indicate the direction of the margins and the cement flow. Note, the Tapered and Chamfer Margin designs direct the cement downwards while the Reverse Margin directs the cement upwards.
B) Margin Position
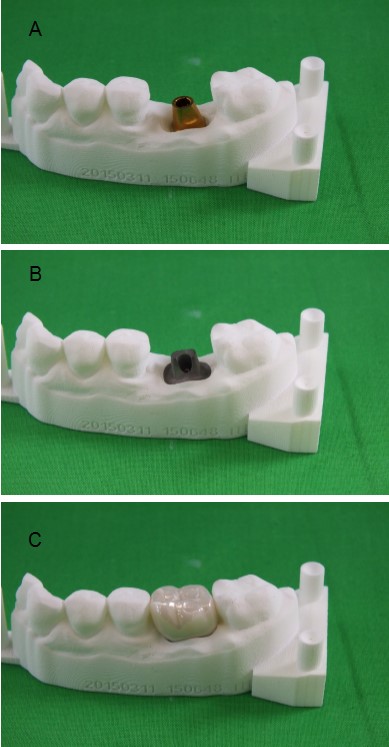
Many authors agree that supra-gingival and equigingival margins are more desirable than subgingival margins for better control of excess cement. Excess cement is easier to see and clean away when margins are above the gingiva. 13 There are a few issues that affect the control of margin position. First, gingival margins are not usually round like machined stock abutments. Figure 2 shows a gold colored round “stock abutment” that is screwed onto its’ implant base.
Fig. 2
The margin on the gold colored abutment is a narrow chamfer pointing downwards, towards the tissues.

Unlike the round shape of such an abutment, gingival margins are usually higher on the mesial and distal, and lower of the facial and lingual. 13 The shape and thickness of gingiva is also highly variable. Figure 3A shows an actual model with a stock abutment screwed in place. It would be very difficult to control the variable gingival contour with such an abutment. Misch 26 and many others have proposed adjusting abutments in the mouth. In the light of recent work by Wilson et al. 5, the creation of metal or zirconia particles in the mouth that may remain in the gingival sulcus give rise to a foreign body response resulting in peri-implantitis. Even if one tried to modify such an abutment, the tissue facing part of the crown would need to be used to create an emergence profile from the round shape of the implant to the shape of the prosthesis. This would make it very difficult to control the important “Gingival Effects” on ejected excess cement.
The good news is, “well designed custom abutments” are available and can be made to allow the clinician to control the emergence profile and the position of the abutment margin with regards to the variable position of the gingiva. Figure 3B shows such a customized abutment with a margin contour that follows the gingival margin and supports the shape of the crown it is designed to retain. Figure 3C shows the crown in place. According to Misch 27, 1 to 1 ½ mm subgingival margin is a good goal for subgingival margins. I would suggest that ½ to 1 mm subgingival might even be better, because it is easier to access during the excess cement cleanup. These recommendations are much better than the 3 mm subgingival margins recommended in the old 2005 edition of Misch’s Book, on Dental Implant Prosthetics.
Fig. 3 A,B&C
A shows a stock abutment in place on a model, It relies on the base of the crown to provide its emergence profile. B shows a customized abutment on the same model. The emergence profile and margin position is created in the abutment. C shows the prosthesis in place.
When the clinician decides to install a prosthesis with subgingival margins, this should stimulate the clinician to make implant selection changes, abutment-prosthesis design changes and procedural changes, to help compensate for increased margin depth and overcome the below-described “Gingival Effects”. Deeper subgingival margins are increasingly difficult to access for the purpose of removing excess cement, even with good cement control.
This is the second step in preventing residual subgingival cement. Use well designed custom abutments to control emergence profile, margin position and keep subgingival margins more accessible for excess cement removal. It is usually very difficult to have margin control and thus optimal cement control with stock abutments and other pre-shaped mass-produced abutments. Well designed custom abutments are important! 19,28
C) Design of the Abutment-Prosthesis Complex
The gingiva adjacent to the implant can have a huge impact on the flow of excess cement during the intra-oral cementation process. The design features of the abutment-prosthesis complex can significantly impact the clinician’s ability to control the flow and cleanup of excess cement. Dr. Emil Svoboda, has designed an “in vitro” gingiva model that allowed him to experiment with and control the “Gingival Effects”. 29-31 The term, “Gingival Effects” was coined by the author to describe the effect of gingiva on the hydraulic movement of excess cement during the intra-oral cementation process. There are four identified effects of the peri-implant gingiva on the flow of cement. They are called the 1) Defection Effect, 2) Eddy Effect, 3) Plunger Effect, and 4) Bellows Effect.
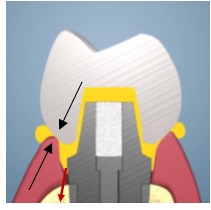
1. The Defection Effect is simply the redirection of excess cement by the gingiva as it comes out from between the prosthesis and the abutment margin. Any downward facing margins will eject cement in a downwards direction and the gingiva will also deflect cement downwards into the tissues. A margin at 90 degrees to the gingiva will be expected to deflect some cement upwards and some downwards. Upward facing margins on the implant or abutment will cause the gingiva to also direct cement upwards, out of the tissues. Figure 4 shows a picture where both the prosthesis and abutment has a chamfer shaped margin (black arrows). The angle of the prosthesis margin tends to direct the cement towards the tissue around the abutment and the gingiva will tend to deflect that cement in the downward tissue direction (red arrows).
Fig. 4
Black arrows point to complimentary margins on prosthesis and abutment. Red arrows show the direction of flow of yellow cement as it is deflected into the tissues.

2. The Eddy Effect was discovered later in the series of experiments conducted by Dr. Svoboda. 31 It occurs when the flow of excess cement out from between the margins is impeded by the flow of the cement out of the gingival crevice while the prosthesis is being seated. It can cause a backflow of cement into the tissues (Fig. 9).
This same experiment with the black tape spacer at the margin, rather than 1 mm under it, extends the width of the margin and prevents cement from breaching the gingival barrier at the margin (Fig. 9).
So, allowing adequate space for the easy escape of excess cement is important, stretching the gingiva to bolster the barrier to excess cement is important, and using low pressure cementation is also important to prevent residual subgingival cement from being expressed into the subgingival spaces. More about cementation pressure below.
The Eddy Effect was named after the Eddies that form at the side of a stream with irregular banks (borders).
3. The Plunger Effect is a huge problem! Figure 5 shows a prosthesis being seated onto the subgingival margin. The wider base of the prosthesis touches and forms a seal with the gingiva prior to the prosthesis being fully seated onto the abutment margin.
Fig. 5
Black arrows indicate the point at which the prosthesis first forms a barrier to movement of excess cement. The red arrows show the direction of flow of yellow cement as it is compressed by the seating of the crown.
This prosthesis-gingival seal (black arrows) can cause a barrier that traps the excess cement already in the subgingival space, while more excess cement still being expressed from the margins of the prosthesis. As the prosthesis continues to be pushed into place, the trapped cement is pressurized. This pressurized excess cement will follow the path of least resistance (red arrows). Some of the excess cement can be propelled deeper into the peri-abutment and/or peri-implant tissue spaces, where it can be difficult to locate and clean away. Figure 6 shows the cement pushing the gingiva laterally (black arrow).
When the backpressure within the tissue space is sufficient (Fig. 7), the cement will overcome the gingiva-prosthesis barrier and again flow up and out from between the gingiva and prosthesis (red arrow). Figure 8 is taken from a video, demonstrating that the Plunger Effect can overcome any of the three tested margin designs. 29,30
This plunger effect is especially bad news for clinicians who use bulky crowns to simulate the emergence profile of a natural tooth while cementing it onto a skinny abutments or implants. Better prepare the patient for surgery to try to locate and remove the excess cement!
4. The Bellows Effect is a phenomenon observed “in vitro” where the simulated gingiva was pushed away by the inferior contour of the crown. This lateral movement of the gingiva increases the space in the peri-abutment environment and thus can create a vacuum between the gingiva and the abutment-implant complex. This vacuum can draw excess cement into the tissue space29, much like a “Bellows” that sucks air into its interior during its filling cycle.
In addition, in a more recent video 30, the author was able to observe that the gingiva (clear Tygon Tubing) ahead of the front of advancing excess cement was pushing the gingiva away from the implant. This action, ahead of the advancing cement could also contribute to the “Bellows Effect” by causing the gingiva to move laterally and thus create a vacuum ahead of its advancing front. This would essentially suck the cement even further into the tissues! The lateral movement of the gingiva depicted in Figures 6 & 7 contributes to the Bellows Effect. The large amount of sub-margin cement shown in Figure 8B also results, in part, by the Bellows Effect. It is best to view the videos pertaining to the Gingival Effects 29,30 and Overcoming the Gingival Effects 31 to understand the Bellows Effects.
Fig. 6
The black arrow indicates the direction of movement of gingiva away from the implant-prosthesis complex. This movement is caused by the crown shape and the flow of excess cement.

Fig. 7
The red arrow indicates the expression of some excess yellow cement once the pressure in the tissues overcomes the barrier of the gingiva-prosthesis seal.

Fig. 8
A Shows 3 aluminum rods with 3 different margin designs that terminate 1 mm below the top of the clear Tygon tubes (gingiva) The black electrical tape was placed 1 mm below the margin and is 0.125 mm thick. It fills the space between the tube and the rod. The black arrows indicate the direction of the margins. B—Depicts the zirconia crowns cemented into place. Note the huge amount of sub-margin cement, regardless of margin design.

The author suspects that the cement plunged into the peri-abutment-implant complex, by the Plunger Effect, may aid in the separation of the gingiva from the abutment-implant complex, and thus facilitate the lateral movement of the gingiva and increase the magnitude of the Bellows Effect. The author suspects, like the Plunger Effect, the Bellows Effect may be especially bad news for clinicians who cement bulky crowns, with margins placed in subgingival locations, onto a skinny abutments or implants. The lateral movement of the gingiva will likely suck cement into the peri-implant tissue space and it will be very difficult to locate and clean away. Get your scalpel ready to help locate and clean away residual excess cement!
In Wadhwani’s book about intra-oral cementation, 32 he has collected images of gross amounts of excess cement still attached to failed dental implants. In the author’s opinion, the Plunger Effect and Bellows Effect could cause such patterns of injected cement with enough cementation pressure and with a relatively wide prosthesis profile that impedes the flow of cement out of the tissue spaces. Look at the gross amount of cement in the tissue spaces of the “in vitro” model. 30,31
Once the above Gingiva Effects were identified, it became a simple matter to make intra-oral cementation safer. The design of the abutment should impede the flow of the cement into the tissue space and the design of the abutment and prosthesis should facilitate the movement of excess cement upwards and out of the tissue space.
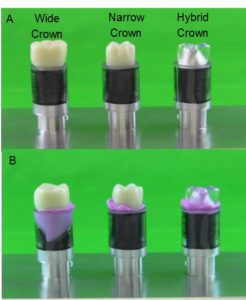
Figure 9 shows the effect of redesigning the prosthesis, to make its’ subgingival profile narrower, so that it does not impede the outflow of excess cement by creating a seal with the adjacent gingiva. Figure 9 also shows that insufficient space to allow the easy flow of cement out of the tissue spaces may still cause some backflow because of the Eddy Effect. The cement moving up and out of the space between the tube and the margin caused some cement to backflow, but it had insufficient pressure to overcome the black tape barrier. 31
Fig. 9
A Shows 3 aluminum rods with Reverse Margins that terminate 1 mm below the top of the clear Tygon tubes (gingiva). The black electrical tape was placed at the margin and is 0.125 mm thick. It fills the space between the tube and the rod. There are 3 crown designs, wide, narrow and hybrid. The narrow is smaller in diameter than the tube, and the hybrid has a narrow part sub-gingival tapering to a wider profile 1/2 mm above the tube. B—Depicts the crowns cemented into place. Note the huge amount of sub-margin cement under the wide crown, while the narrow and hybrid crowns did not have any cement that breached the black tape border.
The margin should be placed so that it is accessible to the clinician for removal of excess cement.
The third step in preventing residual subgingival cement involves design features for the abutment-prosthesis complex that both impede excess cement from going into the sub-margin space and facilitate its movement out of the tissues. Low viscosity or fluid cements are easier to move out of the tissues than thicker cements.
D) Cementation pressure applied by the clinician
Like margin design, implant prosthesis cementation protocol has also been adopted from that used for natural teeth with cements with low compressive strength and complex handling protocols. However, many things have changed over time, including the properties of available cements.
In the past, when using cements with poor handling, compressive strength and solubility characteristics, it was necessary and even desirable to cement crowns with an average seating force of 40 N33 or with even greater forces (up to 600 N) when asking the patient to bite the crown into place. 6,7 These are huge forces! Even low forces of 2.5 N21 seems excessive to the author. In his video 35 he demonstrates an “in vitro cementation of a bridge using a maximum of 0.11 N (110 gms of force). With the low viscosity acrylic based cements available today and larger cement spaces (60-120 microns), it is simply not necessary to use 2500 gms of force to seat a prosthesis.
I am presently using Rely X Ultimate (3M product). I receive no compensation for mentioning this product. Its handling, low viscosity, 12 micron film thickness and high compressive strength (262 MPa), and other characteristics are impressive. There is no need to rush as the cement has a long setting time and is dual cure. The implant prosthesis should already have been tried in and adjusted. The fit of the prosthesis onto the abutments is already confirmed passive. This is easy to achieve with an 80-120 micron cement space. Only minimal force is necessary to “gently finger tap” the prosthesis into place. Then the prosthesis can be held in place for an initial light activated polymerization time. Cleanup is relatively easy. The cementation process, “Prosthesis Installation Technique using the Reverse Margin™ Design and Technique” 22, is described in more detail at www.ReverseMargin.com. 36
Check for updates to this document as it will evolve rapidly through clinician input.
Summary
This article brings to light the ongoing problems with the installation of the assembled “screwed in prosthesis”, as it relates to creating stress-related misfits of the implant-abutment connections and thus rendering them open to bacterial invasion. It also explains how the cement space between the prosthesis and its retainer(s) builds tolerance into the abutment-prosthesis system and thus can compensate for small inaccuracies of prosthesis size and intra-oral retainer position. The cement space is important to allow the clinician to create a passive fit between the prosthesis and its retaining abutment(s). The cement is intended to fill the cement space, retain and support the prosthesis, and also prevent bacterial ingress and growth in the abutment prosthesis interphase.
This article then goes on to identify margin design, margin position and abutment-prosthesis design features that can give the clinician better control of the flow of excess cement and overcome the dreaded “Gingival Effects” that can deflect, redirect, plunge and suck cement into the peri-retainer tissue spaces. Due to the low viscosity of some newer cements and redesign of the abutment-prosthesis complex, the prosthesis does not squeeze the peri-prosthesis gingiva during installation. As a result only very light forces are required to seat the prosthesis on the margin of the abutment. Light cementation forces give the clinician much more control over the flow of excess cement and thus its removal.
Stop causing implant failures! Optimize the foundations of your prosthesis by using a cementation technique that involves custom abutments with Reverse Margin™ design and other features that do not exacerbate the “Gingival Effects”. Then use a cement and technique that allows for optimal cementation control with minimal cementation pressure. For the most current information about preventing residual excess cement, go to www.ReverseMargin.com. 37
Reprinted with permission. Published to www.ReverseMargin.com, August 24, 2015.
References
1. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 26
2. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 28
3. A Systematic Review of Screw- versus Cement-Retained Implant-Supported Fixed Restorations, S. Sherif, H. K. Susarla, T. Kapos, D. Munoz, B. M. Chang, & R. F. Wright, Journal of Prosthodontics 23 (2014) 1–9 .
4. Success or failure of dental implants? A literature review with treatment considerations, J.A.Porter and J.A.von Fraunhofer. AGD Nov/Dec, 2005
5. Foreign Bodies Associated With Peri-implantitis Human Biopsies. Thomas G. Wilson Jr., Pilar Valderrama, Maria Burbano, Jonathan Blansett, Robert Levine, Harvey Kessler., and Danieli C. Rodrigues. J Periodontol 2015;86:9-15
6. Zipprich Micro Movements on Implant Abutment Interfaces part 1. H Zipprich, Published on June 12, 2013, Category People & Blogs, License Standard YouTube License. http://youtu.be/AhsjiYjmTLE
7. Zipprich Micro Movements on Implant Abutment Interfaces part 2. H Zipprich, Published on June 12, 2013, Category People & Blogs, License Standard YouTube License. http://youtu.be/-z5jXFAtfZc
8. Bacterial Leakage Along the Implant-Abutment Interface, Cássio do Nascimento and Rubens Ferreira de Albuquerque Jr. (2011).in Prof. Ilser Turkyilmaz (Ed.), Implant Dentistry–The Most Promising Discipline of Dentistry, ISBN: 978-953-307-481-8, InTech, Available from: http://www.intechopen.com/books/implant-dentistry-the most-promising-discipline-of-dentistry/bacterial-leakage-along-the-implant-abutment-interface
9. Birth of the Reverse MarginTM Design and Order of Magnitude Problem. Emil L.A. Svoboda, Video 3, Published to www.ReverseMargin.com, May 6, 2015.
10. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. Wilson,T.G., J Periodontol 2009;80:1388.
11. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 26:659
12. Alfonso Pineyro DDS, Dentistry’s Dirty Little Secret, AAID Annual Meeting in Oct 26, 2013 Pheonix Arizona,USA.
13. Effect of implant abutment modification on the extrusion of excess cement at the crown-abutment margin for cement-retained implant restorations. C. Wadhwani, A. Piñeyro, T. Hess, H. Zhang, K. Chung. The International journal of oral & maxillofacial implants 11/2011; 26(6):1241-6.
14. Cementation in Dental Implantology. An Evidence Based Guide. Edited by Chandur P.K. Wadhwani. Published by Springer 2015.
15. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 28:738-739
16. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 28:731
17. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 28:739
18. Cementing an Implant Crown: A Novel Measurement System Using Computational Fluid Dynamic Approach. Chandur Wadhwani, Sabine Goodwin and Kwok-Hung Chung. Clinical Implant Dentistry and Related Research, 2014.
19. How Abutment Margin Design Influences Cement Flow: Abutment Selection and Cement Margin Site. Tomas Linkevicius. In Cementation in Dental Implantology. An Evidence Based Guide. Edited by Chandur P.K. Wadhwani. Published by Springer 2015, Ch 6, 98-109
20. The Influence of the cementation margin position on the amount of undetected cement. A prospective clinical study. Tomas Linkevicius et al. Clinical Oral Implants Research. Vol 24,Issue 1, 71-76, Jan 2013.
21. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 26:674
22. Reverse MarginTM, A Great Solution to a Big Problem. Emil L.A. Svoboda, Video 4 Slide Presentation, 2nd edition, Published to www.ReverseMargin.com, May 6, 2015
23. Reverse MarginTM, Protecting Your Foundation. Emil L.A. Svoboda, Animation Video 1, Published to www.ReverseMargin.com, May 6, 2015
24. Reverse MarginTM, Margin Design is Important, Emil L.A. Svoboda, Video 2, Published to www.ReverseMargin.com, May 6, 2015
25. Effects of Margin Design on the Direction of Flow of Excess Cement “in vitro”. Emil L.A. Svoboda PhD, DDS, Video Published to www.ReverseMargin.com. Aug 13, 2015.
26. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 26:666,696
27. Dental Implant Prosthetics, Carl E. Misch, 2nd Edition, Elsevier Mosby, 26:661
28. The Evolution from Stock to Custom Abutments Allows for a Better Prosthesis Design that can Control the Gingival Effects and thus the flow of Excess Cement. Emil L.A. Svoboda PhD, DDS. Published to www.ReverseMargin.com, August 22, 2015
29. Gingival Effects on Intra-oral Prosthesis Cementation. Emil L.A. Svoboda PhD, DDS. Animation Video 6, Published to www.ReverseMargin.com, July 2, 2015.
30. The Gingival Effects can increase the problem of Excess Subgingival Cement. An “in vitro” Study. Emil L.A. Svoboda PhD, DDS, Video Published to www.ReverseMargin.com. Aug 13, 2015.
31. Overcoming the Gingival Effects by Prosthesis Design. An “in vitro” Study. Emil L.A. Svoboda PhD, DDS, Video Published to www.ReverseMargin.com. Aug 17, 2015.
32. Cemented Implant Restorations and the Risk of Peri-implant Disease: Current Status. Chandur P.K. Wadhwani, Thomas G. Wilson Jr. and Kwok-Hung (Albert) Chung. In Cementation in Dental Implantology. An Evidence Based Guide. Edited by Chandur P.K. Wadhwani. Published by Springer 2015, Ch 2, 20-23
33. Effects of finger pressure applied by dentists during cementation of all-ceramic crowns. M.Zortuk, P. Bolpace, K. Kilic, E. Ozdemir, S. Aguloglu. Eur J Dent 2010, Oct; 4(4):383-388.
34. Low force cementation. Wilson PR1. J Dent. 1996 Jul;24 (4):269-73.
35. Super Low Cementation Forces. Emil L.A. Svoboda PhD, DDS, Video Published to www.ReverseMargin.com, July 13, 2015.
36. Prosthesis Installation Technique using the Reverse Margin™ Design and Technique. Emil L.A. Svoboda, Published to www.ReverseMargin.com. June 16, 2015.
37. Stop causing Implant Failures! Emil L.A. Svoboda, Video 5, Slide Presentation, Published to www.ReverseMargin.com, May 9, 2015
About the Author
Dr. Emil L.A. Svoboda earned his BSC (1974), PhD (1978) and DDS (1982) at the University of Toronto. He then earned his Fellowship with the Academy of General Dentistry, Fellowship with the American Academy of Implant Dentistry and is a Diplomate of the American board of Oral Implantlogy/Implant Dentistry. More recently he was presented with the “Honoured Status” by the American Academy of Implant Dentistry and he was given the Award of Merit by the Ontario Dental Association. Dr. Svoboda has lectured about implant dentistry worldwide and continues to educate dentists about implant surgery and prosthetics.












