Abstract
The dental profession represents a significant player in the future trajectory of the current opioid crisis. Exercising sound clinical judgement and adhering to evidence-based principles of pain management are essential. The aim of the present report is to review the current landscape of the opioid crisis and present evidence-based acute pain management strategies in dentistry.
The Current Opioid Crisis
Prescribing opioids in the management of acute and chronic pain is a common practice in North America and across Ontario.1-3 In fact, Canadians are the second largest per-capita users of prescription opioids worldwide.4 The opioid crisis in Canada presents a major public health concern that has been attributed to the widespread use of prescription opioids to treat non-cancer pain.5,6 One in eight patients in Ontario are treated with opioids to manage pain.1 Higher rates of opioid administration have been closely associated with an increase in adverse events, including motor vehicle accidents, the development of opioid use disorder, depression, opioid overdose, and death.7-10 The rate of opioid-related deaths in Ontario has tripled between 2000 and 2015. In 2015, one in every 133 deaths in Ontario was opioid-related, with this burden being the largest among young adults aged 15 to 24 years where one in every six deaths was opioid-related.11 In 2016, one in three opioid-related deaths occurred among people with an active opioid prescription and more than 75% of opioid-related deaths had an opioid dispensed within the past three years.12 Thus, prescription opioids are a significant contributor to the ongoing opioid crisis in North America.
Health Quality Ontario (HQO) reported that dentists prescribe 16.6% of all opioids in Ontario when compared to other healthcare professionals.2 Following family physicians, dentists represent the second largest group of opioid prescribers in Ontario.2 Nearly 75% of dentists’ opioid prescriptions are new starts, which includes individuals who have not filled an opioid prescription in the past six months or those who are being exposed to opioids for the first time. It has been demonstrated that dentists represent a significant source of initial exposure to opioids.13 In 2016, Hydromorphone contributed to 0.7% of opioid new starts prescribed by dentists while Tramadol accounted for 3.2% of the total new starts by dentists. Codeine combination prescriptions, such as Tylenol 3, were responsible for 80% of all new starts, a trend that remains consistent.2 This is concerning, as opioid use disorder often occurs from an initial chance exposure to prescription opioids.5, 14 In fact, prescription opioid use before 12th grade has been shown to significantly predict future opioid misuse following high school.15 It is evident that dental professionals have an important role to play in the ongoing opioid crisis, and that practicing evidence-based pain management strategies can positively impact the future trajectory of this crisis.
Several policies and legislations have been implemented in the healthcare sector in efforts to combat the opioid crisis. The Narcotic Safety and Awareness Act (NSAA) is provincial legislation that was implemented in 2011, outlining certain requirements that the patient, prescriber, and dispenser must follow when accessing monitored drugs. A monitored drug is considered a controlled substance under the Controlled Drugs and Substances Act (CDSA) and other opioid medications not listed in the CDSA, such as Tramadol and Tapentadol containing products. The aim of this act is to reduce the abuse, misuse, and diversion of monitored drugs through the promotion of appropriate prescribing and dispensing practices.16 Safeguarding our Communities Act was implemented at the provincial level in 2016 and mandates a fentanyl patch-for-patch exchange system. In order for dispensers to release another fentanyl patch to a patient, the previously used patch must be returned.17 Further, the narcotic monitoring system (NMS) is a central database to review drug prescribing and dispensing activities in the community healthcare sector. This system contains real-time Drug Utilization Review (DUR) that will be performed each time a dispensing record is submitted to the NMS by a pharmacy. If issues such as double doctoring or polypharmacy are detected, the NMS will notify the pharmacy in real-time as the prescription is being dispensed. These safeguards indicate a growing effort to resolve the opioid crisis across all healthcare professions. In dentistry, sound prescribing practices can aid this overall effort to combat the opioid crisis.
Acute Pain in Dentistry
Background
In the 1970s, Steve Cooper and William Beaver developed and validated a third molar removal model that compared the efficacy of different oral analgesics and a placebo control arm.18 The third molar removal model studied the subjective experience of pain effectively for several reasons. Third molar removal is a commonly performed procedure that causes similar surgical trauma for the majority of patients, making the pain experience relatively ubiquitous. This allows the sample size to be large while minimizing individual variations in the pain experienced by each patient. The procedure is often performed in both the left and right sides of a patient’s mouth, allowing split-mouth designs to be utilized to further minimize subjective pain experience. Additionally, third molar removal is performed in young healthy adults which helps rule out several confounding variables in an experimental setting. In their hallmark study, Cooper and Beaver demonstrated that Ibuprofen (400mg) was more efficacious than Codeine (60mg), Aspirin (650mg), or both analgesics in combination.18 Clinical trials have consistently demonstrated that non-steroidal anti-inflammatory drugs (NSAIDs), administered as sole agents, are effective in the management of post-operative dental pain.19-22 Moore et al. (2018) demonstrated that 600mg of Ibuprofen resulted in the highest proportion of patients who experienced pain relief, followed by 400mg Ibuprofen combined with 1000mg Acetaminophen.23 Understanding the relevant findings in the literature, general prescription principles, and treatment algorithms or guidelines will aid clinicians in making informed pain management decisions.
General Principles
General principles must be adhered to for adequate pain management. It is prudent to never administer a drug without an indication. Clinicians must balance the benefits and risks, and only continue if the balance is favourable. Moreover, all drugs have the potential to cause drug interactions and adverse drug reactions. The first alternative to pain management should include non-pharmacological methods. If possible, the source of the pain should be eliminated directly. In dental practice, this includes root canal therapy, extraction, or incision and drainage. Analgesics help to mitigate pain, but do not eliminate pain. For acute post-operative dental pain, the use of analgesics should be individualized to each patient based on their medical history and level of pain experienced. Other factors to consider when prescribing include allergies, intolerances or prior adverse drug reactions, pregnancy and lactation, as well as hepatic and renal function. Dentists should avoid prolonged use of any analgesic agent. The duration of post-operative dental pain is typically limited to three to five days.
Management of Acute Dental Pain
An algorithm has been established in order to exercise best prescribing practices.24 For the management of acute dental pain, opioids are seen as third-line therapy following Acetaminophen and NSAIDs, and should only be considered by the clinician once the dosages of the non-opioids have been maximized.
Acetaminophen is the first drug of choice and should always be considered from a risk-benefit standpoint, particularly when pain severity is expected to be mild to moderate. An adult dose of Acetaminophen is initially considered (Table 1; 500-1,000mg q4h, maximum 4g/day). If 1,000mg of Acetaminophen is determined to be insufficient for effective analgesia, and the patient has no contraindications to NSAIDs (Table 2), the next step is to recommend an NSAID (Ibuprofen 200-400mg q4-6h, as needed for pain). However, if the patient has a contraindication to NSAIDs (Table 2), adding codeine to Acetaminophen may be indicated. If the pain remains severe and more analgesia is required following the initial NSAID prescription, the maximum dose of NSAID is prescribed (Ibuprofen 600mg), or a combination of 1,000mg of Acetaminophen and 400mg Ibuprofen is considered.25,26 Alternatively, adding an opioid (30-60mg codeine or 5-10mg oxycodone combination) to the NSAID or Acetaminophen may also be considered. The clinician must optimize the dose and the frequency to confirm that compliance is not the underlying reason for inadequate pain relief prior to adding an opioid.
Table 1

Note. NSAIDs are listed in decreasing selectivity for COX-2/increasing selectivity for COX-1 (with the exception of Floctafenine/Idarac)
Table 2

Adapted from Haas 2002.24 Therapeutic effects of each analgesic agent are numbered. The primary therapeutic effects utilized in dentistry are analgesia and anti-inflammation.
If the clinician anticipates severe post-operative pain prior to the procedure based on the nature of the procedure or the patient’s pain threshold, a pre-operative dose, loading dose, or around-the-clock dosing of NSAIDs may be indicated. Pre-operative NSAID dosing has been demonstrated to decrease the severity and onset of acute post-operative dental pain.27 However, loading doses are generally favoured, as pre-operative NSAIDs are best avoided when bleeding concerns exist.24 A loading dose is simply two tablets stat, immediately after the procedure, followed by one tablet every 4-6 hours as needed for pain. The purpose is to reach steady state levels of NSAIDs prior to the dissipation of the local anaesthesia that was initially administered. An alternative dosing regimen for severe pain is around-the-clock for the first one to three days, which aids in managing pain prior to its onset. Long-duration local anaesthetics such as 0.5% Bupivacaine can be utilized at the end of procedures to provide prolonged post-operative analgesia.28 This approach results in a reduced need for post-operative opioids.29 Clinical judgement is required in each clinical scenario to determine if a pre-operative dose, loading dose, or around-the-clock regimen of NSAIDs is appropriate.
There are different reasons one may experience pain relief following the administration of an analgesic. It is possible that the drug was efficacious and responsible for the alleviation of pain. However, the temporal relationship between the administration of the drug and the dissipation of pain may give patients reason to believe the drug was effective when, in fact, the pain would have abated without any analgesics to begin with. Another reason for pain relief is the placebo effect whereby no pharmacological effect exists. It is purely a psychological effect that accounts for the alleviation of pain. For example, patients often assume that more expensive drugs and prescribed drugs inherently have greater efficacy. Clinicians may prescribe 400mg of Ibuprofen rather than instructing the patient to purchase it over-the-counter, as it may result in a psychological benefit to pain relief.
Acetaminophen
Acetaminophen is the first alternative considered for post-operative dental pain in elderly, adult, and paediatric patients due to its favourable risk-benefit profile.24 Acetaminophen is effective for mild to moderate pain, providing both analgesic and antipyretic properties without the gastric side effects of NSAIDs.24 It is important to inform patients to avoid other Acetaminophen-containing preparations such as Midol© or NyQuil©, as this may cause inadvertent overdose. Acetaminophen toxicity has been demonstrated when daily doses of 4,000mg are exceeded.30 Hepatotoxicity has also been reported in febrile pediatric patients as a result of several dose miscalculations and subsequent administration by the parents.31
Approximately 95% of Acetaminophen detoxification is accomplished by glucuronidation and sulfation, with these metabolites undergoing rapid renal elimination (Fig. 1). Under normal circumstances, a small percentage (5%) of Acetaminophen is oxidized by cytochrome P450 enzymes (CYP2E1) into N-acetyl-p-benzoquinoneimine (NAPQI), a potentially toxic metabolite of Acetaminophen. This does not lead to hepatotoxicity in healthy individuals, as NAPQI is conjugated and inactivated by glutathione.32 When excessive amounts of Acetaminophen are consumed, the conjugation pathways (glucuronidation, sulfation, glutathionylation) become saturated and NAPQI accumulates, leading to significant hepatotoxicity.33-35 N-acetylcysteine is the antidote of choice for Acetaminophen toxicity. It must be administered prior to the clinical symptoms of overdose, typically within the first 16 hours of consumption.22,35 It is important to understand that so long as correct dosing instructions are provided by the clinician, the likelihood of adverse reactions is generally low.36
Fig. 1

Non-Steroidal Anti-Inflammatory Drugs
NSAIDs inhibit the action of cyclooxygenase-1 and cyclooxygenase-2 enzymes (COX-1 and COX-2) in the bloodstream, decreasing the production of prostaglandins, thromboxanes, and prostacyclin (Fig. 2). Aspirin irreversibly acetylates COX-1 and COX-2, resulting in decreased levels of prostaglandins and thromboxane A2.37 COX-1 is constitutively expressed and produces prostaglandins, which provide gastric protection, regulate renal blood flow, and stimulate uterine contraction. COX-1 also aids in hemostasis by producing thromboxane, a potent vasoconstrictor and initiator of platelet aggregation. COX-2 is induced during dental procedures that inflict cellular damage, augmenting the production of prostaglandins that mediate pain and inflammation.
Fig. 2
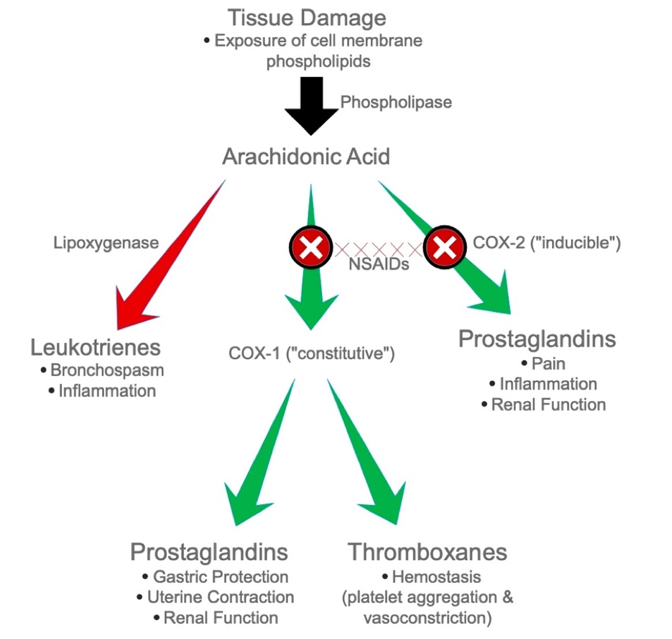
NSAIDs produce both therapeutic and adverse effects when COX-1 and COX-2 are blocked. In contrast to Acetaminophen, NSAIDs have several drug interactions such as reducing the efficacy of antihypertensives, potential toxicity when combined with Lithium and Methotrexate, and increased bleeding with selective serotonin reuptake inhibitors (SSRIs) and other anti-platelets, all of which require additional management considerations (Table 3). NSAIDs are indicated for their analgesic, anti-inflammatory, and anti-pyretic properties. Adverse effects and contraindications of NSAIDs are listed in Table 2. By inhibiting COX-1, the cytoprotective effect of prostaglandins is lost, and the patient is more susceptible to dyspepsia, which may lead to serious side effects such as gastric mucosal damage and bleeding. In fact, 16,500 deaths in the United States are attributed to gastric bleeding induced by NSAIDs.38 Further, decreased thromboxane production leads to impaired hemostasis, rendering the patient susceptible to increased bleeding. Patients with a history of renal disease may develop renal impairment following NSAID use. A patient may have an Aspirin (ASA) allergy, or an allergic-like reaction, otherwise known as an anaphylactoid reaction. In this instance, arachidonic acid is shunted towards the leukotriene pathway, causing bronchospasm and other symptoms mimicking anaphylaxis that can be extremely severe and life threatening (Fig. 2). The adverse effects of NSAIDs are often dose and duration dependent. Thus, it is recommended to utilize the lowest effective dose for the shortest possible duration to eliminate acute pain.39
Table 3

Note. This list of drug interactions is not complete
Opioids
The primary indication for the use of opioids in dentistry is for their analgesic properties. Opioids have a profound ability to raise pain threshold and alter pain reactions, both of which contribute to incredibly effective analgesia. Opioids have the potential to give patients a euphoric feeling, which may contribute to the analgesic effect. However, many other pharmacological effects may result following administration, including respiratory depression, sedation, hypo- and hypertension, nausea, vomiting, constipation, and urinary retention (Table 2). The development of tolerance, physical dependence, and addiction require special consideration. Tolerance may develop rapidly and can be seen in patients following weeks of repeated intake, where higher doses are required in order to achieve the same analgesic effect. A state of physical dependence results when an individual requires a drug for the maintenance of normal homeostasis. After discontinuation of the drug, the patient experiences withdrawal symptoms such as rhinorrhea, piloerection, abdominal cramps, diarrhea, restlessness, shivering, excessive perspiration, increased blood pressure and insomnia. Studies have indicated that patients who take opioids for longer than seven days are more likely to become physically dependent.40 Drug abuse is the inappropriate use of a drug for non-medical reasons and can lead to behavioural and physical dependence if sustained. Above all, an opioid addiction may develop.41 In addiction, the patient continues to crave opioids regardless of their known negative consequences and adverse effects. Clinicians must be aware of the signs that indicate aberrant drug related behaviours. Patients who request higher doses, complete analgesic regimens far too early, “lose their pills frequently”, and alter the route of delivery should be monitored closely by the clinician. The extensiveness and severity of these side effects elicited by opioids has led to the current opioid crisis.
Although opioid analgesics are not first-line agents for managing acute pain in dentistry, there are situations in which they are indicated and necessary. Three opioids that are most commonly prescribed in dentistry are Codeine, Oxycodone, and Hydromorphone (Table 4). Prescribing opioids should only be considered when in combination with Acetaminophen or an NSAID, at specified doses and intervals (Table 4). When an opioid is indicated, Codeine is the first opioid to consider. Formulations that combine Acetaminophen with Codeine are commonly utilized (Table 5). If codeine is determined to be insufficient, Oxycodone is the next opioid to consider. Oxycodone is combined with Acetaminophen (Percocet) or ASA (Percodan), and is deemed to be more effective than Codeine. Hydromorphone plays an important role for patients with severe chronic pain, and thus is rarely used in a dental setting to manage acute pain. Combination drugs have become popular due to ease of administration, but do not follow the basic principle of maximizing non-opioids prior to administering opioids. For example, the Tylenol 4 preparation contains 300mg of Acetaminophen and 60mg of codeine (Table 5), maximizing only the opioid dose with a sub-therapeutic amount of Acetaminophen.
Table 4

Table 5

As it has become a common practice in dentistry to routinely prescribe opioid combinations to manage post-operative pain. The Royal College of Dental Surgeons of Ontario (RCDSO) outlined factors that may be contributing to the over-prescription of opioids.42 Dentists may be overprescribing opioids out of habit, convenience, or simply lack of knowledge regarding the efficacy of non-opioid analgesics. Patients may demand or expect the dentist to prescribe opioids, and it has been suggested that dentists are prescribing opioids in situations when they are not indicated in hopes to avoid conflicts with their patients. The reasons for over-prescribing outlined by the RCDSO can be avoided with measures aimed at educating both clinicians and patients alike. The RCDSO has indicated that only a minority of the most severe clinical cases require opioid analgesia.42
Guidelines for Opioid Management
In 2015, RCDSO published an opioid prescribing guideline that outlines the management of pain in dentistry. Sequential steps need to be taken in order to minimize prescription opioid abuse. Prior to prescribing an opioid, is it prudent to consider whether the patient’s pain is well documented, if an opioid is currently being taken, if signs of substance misuse, abuse, and/or diversion may be inferred from their medical history, and to weigh the benefits and risks of prescribing an opioid.42 If an opioid is to be prescribed, the recommended maximum number of opioid tablets that should be dispensed is illustrated in Table 6.
Table 6

If the patient returns complaining of unmanaged pain following the first prescription of an opioid, a clinical reassessment, confirmation of diagnosis, and considerations regarding the effectiveness of the opioid need to be accounted for prior to giving an increased dose of the opioid.
If the patient returns for a third appointment and is presenting with unmanaged pain, reassessment and confirmation of diagnosis must be repeated. At this point, a possible third prescription may be issued. However, it is crucial to advise the patient that no further opioid prescriptions will be issued. Interprofessional collaboration must be utilized to address the pain management adequately and to prevent the potential for misuse, abuse, and/or diversion.
Special Populations
It is essential to individualize pharmacological regimens for all patients, particularly for patient populations that require special attention. A modification in the prescribed dose and frequency is often necessary for paediatric and elderly patients, as well as for pregnant or breastfeeding mothers.
Paediatric Patients
In paediatrics, Acetaminophen is the first analgesic to consider, given at a dose of 10-15mg/kg q4-6h with a maximum dose of 75mg/kg. If further pain management is needed, Ibuprofen may be given as a liquid formulation at a dose of 10mg/kg q6-8h for children aged 2-12, and 200-400mg q4h for children over 12 years old, with a daily maximum of 1,200mg. ASA is contraindicated in paediatric patients due to their potential to cause Reye’s Syndrome 42 Health Canada and the FDA do not recommend opioid prescriptions in patients aged 12 years or younger.
Elderly Patients
Canadians over the age of 65 make up 15% of the patient population, and account for over 30% of pharmaceutical prescriptions. It is projected that 25% of Canadians will be over the age of 65 in 2030.44 Dentists must understand the pharmacodynamic and pharmacokinetic changes in elderly patients in order to provide safe and effective pharmacological pain management. When possible, eliminating the source of pain using non-pharmacologic measures is prudent. If pharmacotherapy is required for pain management, Acetaminophen is the analgesic of choice. There are relatively minimal drug interactions with Acetaminophen, which is particularly important in elderly patients who may be currently taking multiple medications.44 NSAIDs adversely effects the gastrointestinal tract by causing gastric bleeding, erosion, and ulceration, all of which may be life threatening in the geriatric population. When an NSAID is indicated, the dose and duration should be reduced.44 In the case where an elderly patient with a history of gastric bleeding is required to take an NSAID, the clinician should consider either administering the NSAID concurrently with a prostaglandin analog such as misoprostol, or prescribing a COX-2 inhibitor such as celecoxib, which has reduced gastrointestinal effects. Opioids are used as a last resort and should be avoided as elderly patients are more susceptible to their adverse effects. The depth and duration of the analgesic effect of opioids is significantly increased in elderly patients. If an opioid is indicated, the dose should be prescribed at half of the recommended adult dose.44
Pregnancy and Lactation
For a pregnant patient, the first recommendation is to identify and eliminate the source of pain when possible. During pregnancy, patients are undergoing physiological changes that alter the pharmacokinetics of drugs, making some analgesics more appropriate to prescribe than others. When an analgesic is indicated, Acetaminophen is the drug of choice during all three semesters of pregnancy. The use of NSAIDs is less favourable and is a contraindication in the third trimester as it causes increased bleeding, prolonged labour, and premature closure of the ductus arteriosus. NSAIDs may be used cautiously in the 1st and 2nd trimester, using the lowest effective dose for the shortest possible time. In general, opioids should be avoided during pregnancy.45 However, if a pregnant patient is experiencing moderate to severe pain that cannot be managed by Acetaminophen, an opioid may be used in the 2nd and 3rd trimester at a low dose for short duration.
In the majority of instances, there is a negligible effect of most drugs on a nursing infant. As with pregnancy, the drug of choice during lactation is Acetaminophen. ASA may interfere with the infant’s platelet function leading to increase bleeding and thus should be avoided. Opioids should also be avoided as they can cause extreme drowsiness in the nursing infant.45
Conclusion
Prescription opioids continue to contribute to the ongoing opioid crisis. As common prescribers of opioids, dentists have a duty to help prevent its diversion, misuse, and abuse. Proper management of acute post-operative dental pain grounded in evidence-based principles is critical. Dentists are responsible for adhering to guidelines and individualizing post-operative analgesic care, while being cognisant of the fact that every opioid prescription has far reaching implications, affecting the current and future landscape of the opioid epidemic.
Oral Health welcomes this original article.
References
- Gomes, T., Pasricha, S., Martins, D. & Greaves, S., et al. Behind the Prescriptions: A snapshot of opioid use across all Ontarians. 1–22 (2017). doi:10.31027/ODPRN.2017.04.
- Health Quality Ontario. Starting on Opioids, Health Quality Ontario Specialized Report. (2018).
- Clark, D. J. & Schumacher, M. A. America’s Opioid Epidemic: Supply and Demand Considerations. Anesth. Analg. 125, 1667–1674 (2017).
- Fischer, B. & Argento, E. Prescription opioid related misuse, harms, diversion and interventions in Canada: a review. Pain physician 15, (2012).
- Dhalla, I. A., Persaud, N. & Juurlink, D. N. Facing up to the prescription opioid crisis. BMJ (Online) 343, (2011).
- Boudreau, D. et al. Trends in long-term opioid therapy for chronic non-cancer pain. Pharmacoepidemiol. Drug Saf. 18, 1166–1175 (2009).
- Bohnert, A. S. B. et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA – J. Am. Med. Assoc. 305, 1315–1321 (2011).
- Gomes, T., Mamdani, M. M., Dhalla, I. A., Michael Paterson, J. & Juurlink, D. N. Opioid dose and drug-related mortality in patients with nonmalignant pain. Arch. Intern. Med. 171, 686–691 (2011).
- Scherrer, J. F. et al. The influence of prescription opioid use duration and dose on development of treatment resistant depression. Prev. Med. (Baltim). 91, 110–116 (2016).
- Engeland, A., Skurtveit, S. & Mørland, J. Risk of Road Traffic Accidents Associated With the Prescription of Drugs: A Registry-Based Cohort Study. Ann. Epidemiol. 17, 597–602 (2007).
- Gomes, T. et al. Measuring the Burden of Opioid-related Mortality in Ontario, Canada. Journal of addiction medicine 12, 418–419 (2018).
- Gomes, T. et al. Contributions of prescribed and non-prescribed opioids to opioid related deaths: Population based cohort study in Ontario, Canada. BMJ 362, (2018).
- Pasricha, S. V et al. Clinical indications associated with opioid initiation for pain management in Ontario, Canada: A population-based cohort study. Pain 159, 1562–1568 (2018).
- Chou, R. et al. Opioids for Chronic Noncancer Pain: Prediction and Identification of Aberrant Drug-Related Behaviors: A Review of the Evidence for an American Pain Society and American Academy of Pain Medicine Clinical Practice Guideline. J. Pain 10, (2009).
- Miech, R., Johnston, L., O’Malley, P. M., Keyes, K. M. & Heard, K. Prescription opioids in adolescence and future opioid misuse. Pediatrics 136, e1169–e1177 (2015).
- Ontario’s Narcotics Strategy – The Narcotics Safety and Awareness Act – MOHLTC. Available at: http://www.health.gov.on.ca/en/public/programs/drugs/ons/ons_legislation.aspx. (Accessed: 11th January 2020)
- Ontario College of Pharmacist’s. Patch-For-Patch Fentanyl Return Program: Fact Sheet. (2018). Available at: https://www.ocpinfo.com/regulations-standards/practice-policies-guidelines/Patch_For_Patch_Fentanyl_Return_Fact_Sheet/. (Accessed: 11th January 2020)
- Cooper, S. A. & Beaver, W. T. A model to evaluate mild analgesics in oral surgery outpatients. Clin. Pharmacol. Ther. 20, 241–250 (1976).
- Dionne, R. A. & Berthold, C. W. Therapeutic uses of non-steroidal anti-inflammatory drugs in dentistry. Critical Reviews in Oral Biology and Medicine 12, 315–330 (2001).
- Ahmad, N. et al. The efficacy of nonopioid analgesics for postoperative dental pain: a meta-analysis. Anesth. Prog. 44, 119–126 (1997).
- Dionne, R. A. & Gordon, S. M. Nonsteroidal anti-inflammatory drugs for acute pain control. Dental Clinics of North America 38, 645–667 (1994).
- Hersh, E. V., Moore, P. A. & Ross, G. L. Over-the-counter analgesics and antipyretics: A critical assessment. Clin. Ther. 22, 500–548 (2000).
- Moore, P. A. et al. Benefits and harms associated with analgesic medications used in the management of acute dental pain: An overview of systematic reviews. J. Am. Dent. Assoc. 149, 256-265.e3 (2018).
- Haas, D. A. An update on analgesics for the management of acute postoperative dental pain. J. Can. Dent. Assoc. 68, 476–482 (2002).
- Moore, R. A., Derry, S., McQuay, H. J. & Wiffen, P. J. Single dose oral analgesics for acute postoperative pain in adults. Cochrane database Syst. Rev. CD008659 (2011). doi:10.1002/14651858.CD008659.pub2
- Moore, P. A. & Hersh, E. V. Combining ibuprofen and acetaminophen for acute pain management after third-molar extractions: Translating clinical research to dental practice. J. Am. Dent. Assoc. 144, 898–908 (2013).
- Hersh, E. V. et al. Prescribing recommendations for the treatment of acute pain in dentistry. Compend. Contin. Educ. Dent. 32, (2011).
- Danielsson, K., Evers, H. & Nordenram, A. Long-acting local anesthetics in oral surgery: an experimental evaluation of bupivacaine and etidocaine for oral infiltration anesthesia. Anesth. Prog. 32, 65–8
- Jackson, D. L., Moore, P. A. & Hargreaves, K. M. Preoperative nonsteroidal anti-inflammatory medication for the prevention of postoperative dental pain. J. Am. Dent. Assoc. 119, 641–647 (1989).
- Guggenheimer, J. & Moore, P. A. The therapeutic applications of and risks associated with acetaminophen use: A review and update. Journal of the American Dental Association 142, 38–44 (2011).
- Rivera-Penera, T. et al. Outcome of acetaminophen overdose in pediatric patients and factors contributing to hepatotoxicity. J. Pediatr. 130, 300–304 (1997).
- Haas, D. A. Adverse drug interactions in dental practice: Interactions associated with analgesics: Part III in a series. J. Am. Dent. Assoc. 130, 397–407 (1999).
- Turkoski, B. B. Acetaminophen: Old friend – New rules. Orthop. Nurs. 29, 41–43 (2010).
- James, L. P., Mayeux, P. R. & Hinson, J. A. Acetaminophen-induced hepatotoxicity. Drug Metabolism and Disposition 31, 1499–1506 (2003).
- Rumack, B. H. Acetaminophen hepatotoxicity: The first 35 years. in Journal of Toxicology – Clinical Toxicology 40, 3–20 (2002).
- Ouanounou, A., Ng, K. & Chaban, P. Adverse drug reactions in dentistry. International Dental Journal idj.12540 (2020). doi:10.1111/idj.12540
- Vane, J. R. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat. New Biol. 231, 232–235 (1971).
- Wolfe, M. M., Lichtenstein, D. R. & Singh, G. Gastrointestinal toxicity of nonsteroidal antiinflammatory drugs. New England Journal of Medicine 340, 1888–1899 (1999).
- Bally, M. et al. Risk of acute myocardial infarction with NSAIDs in real world use: Bayesian meta-analysis of individual patient data. BMJ 357, (2017).
- Dowell, D., Haegerich, T. M. & Chou, R. CDC guideline for prescribing opioids for chronic pain-United States, 2016. JAMA – J. Am. Med. Assoc. 315, 1624–1645 (2016).
- Olsen, Y. The CDC guideline on opioid prescribing: Rising to the challenge. JAMA – Journal of the American Medical Association 315, 1577–1579 (2016).
- Royal College of Dental Surgeons of Ontario. Guidelines – The Role of Opioids in the Management of Acute and Chronic Pain in Dental Practice. (2015).
- Ouanounou, A. & Haas, D. A. Pharmacotherapy for the Elderly Dental Patient. J Can Dent Assoc 80 (2015).
- Ouanounou, A. & Haas, D.A. Drug Therapy during Pregnancy: Implications for the Dental Practice. British Denta journal. 220(8):413-417 (2016).
About the Authors
 Mr. Andrew Lombardi is a 3rd year dental student at the Faculty of Dentistry, University of Toronto.
Mr. Andrew Lombardi is a 3rd year dental student at the Faculty of Dentistry, University of Toronto.
 Ms. Sarah Lam is a 3rd year dental student at the Faculty of Dentistry, University of Toronto.
Ms. Sarah Lam is a 3rd year dental student at the Faculty of Dentistry, University of Toronto.
 Dr. Aviv Ouanounou† is an assistant professor of Pharmacology and Preventive Dentistry at the Faculty of Dentistry, University of Toronto, and is also a clinical instructor and Treatment Plan Coordinator in the clinics. Dr. Ouanounou is the recipient of the 2014-2015 prestigious Dr. Bruce Hord Master Teacher Award for excellence in teaching and is the 2018-2019 Recipient of the W.W. Wood Award for Excellence in Dental Education Dr. Ouanounou is a Fellow of the International College of Dentists, the American College of Dentists and the Pierre Fouchard Academy. He is a member of the American Academy of Pain Management and the American College of Clinical Pharmacology. Dr. Aviv Ouanounou is the corresponding author for this article and he can be reached at aviv.ouanounou@dentistry.utoronto.ca.
Dr. Aviv Ouanounou† is an assistant professor of Pharmacology and Preventive Dentistry at the Faculty of Dentistry, University of Toronto, and is also a clinical instructor and Treatment Plan Coordinator in the clinics. Dr. Ouanounou is the recipient of the 2014-2015 prestigious Dr. Bruce Hord Master Teacher Award for excellence in teaching and is the 2018-2019 Recipient of the W.W. Wood Award for Excellence in Dental Education Dr. Ouanounou is a Fellow of the International College of Dentists, the American College of Dentists and the Pierre Fouchard Academy. He is a member of the American Academy of Pain Management and the American College of Clinical Pharmacology. Dr. Aviv Ouanounou is the corresponding author for this article and he can be reached at aviv.ouanounou@dentistry.utoronto.ca.












