Osteochondroma (OC) is also known as osteocartilaginous exostosis. It arises from the bone cortex as an exophytic lesion with a hyaline cartilaginous cap. It is one of the most common benign tumors of the axial skeleton. It accounts for approximately one-third of benign bony lesions. OC may arise in any bone that develops from endochondral ossification and it is rare in maxillofacial region as most of the craniofacial skeleton develops from intramembranous ossification. Embryonic development of temporomandibular joint by endochondral ossification predisposes the coronoid and condylar process. The other reported sites are mandibular symphysis, body of mandible, in the soft tissues at the angle of the mandible, maxillary sinus and posterior maxilla. OC is usually seen in younger individuals with male predominance. OC may present in a solitary fashion or as multiple OCs seen in autosomal dominant syndrome known as osteochondromatosis.1
Report of a case:
A 32 year old female presented to the senior author with a 3 year history of progressively worsening malocclusion and associated left TMJ pain and limited interincisal opening. She complained of severe pain being 8/10 on the visual analogue scale. Her medical history was otherwise noncontributory.
The clinical examination confirmed facial asymmetry and left TMJ pain with palpation. She had a class III malocclusion with severe open bite and asymmetry and deviation of the mandible to the right. Her interincisal opening was moderately limited to 32 mm with deviation to the left. (Figs. 1-4)
Fig. 1

Fig. 2

Fig. 3

Fig. 4

Radiographic investigation included panoramic films and CT (Figs. 5,6). These showed a large exophytic growth associated with the left mandibular condyle. The mass was lobulated and extended into the infratemporal fossa, causing compression deformity of the posterior wall of the maxillary sinus. It was benign looking. The radiographic appearance was in keeping with an actively growing osteochondroma of the left TMJ. The medial extent of the lesion suggested potentially dangerous proximity to the large arteries of the base of skull and medial mandibular space (internal maxillary artery, middle meningeal artery, accessory meningeal artery).
Fig. 5

Fig. 6

The patient was advised to have the tumour removed. Simultaneous correction of the malocclusion (orthognathic surgery) would be achieved with a contralateral sagittal split osteotomy of the mandible. The left condyle would be reconstructed with a custom alloplastic total joint prosthesis designed to fit the new occlusion. The prosthesis is virtually designed and manufactured with CAD-CAM technology. The occlusal alignment splint is virtually designed and manufactured with 3D printing.
The surgical plan also called for a left zygomatic arch osteotomy to provide access to the infratemporal fossa for delivery of the large tumour and control of potentially severe associated bleeding. A portion of the affected arch would be incorporated into the custom glenoid fossa prosthesis.
Orthodontic treatment was recommended to coordinate the dentition to optimize the occlusal correction.
Collaboration with Otolaryngology – Head and Neck Oncology Surgery was recommended to isolate and protect the facial nerve. Their experience with base of skull surgery and oncologic neck surgery would provide optimal team capability should a severe bleed occur during surgery. This would include pre-emptive exposure of the external carotid artery should ligation become necessary in the event of a severe bleed intraoperatively.
Collaboration with Interventional Neuroradiology was recommended to acquire CT angiography to assess the proximity of the arteries near the tumour and to perform arterial embolization, blocking the vessels immediately prior to surgery. This is done by feeding a cannula up through the femoral artery to the carotid artery system and then into the internal maxillary arterial system. Coils and gelfoam plugs are then inserted through the cannula, blocking these vessels.
The patient was prepared orthodontically. She then had a high-resolution medical grade 3D CT scan. The data was uploaded to 3D Systems (Denver Colorado). A web meeting was carried out with the primary surgeon (DJP), biomedical engineers and engineers from Zimmer Biomet (manufacturer of the prostheses). The prosthesis was thus designed and approved for manufacture. The patient was also tested for chromium, cobalt, titanium and polyethylene allergies to rule out metal allergies. (Figs. 14, 15, 16)
Fig. 14
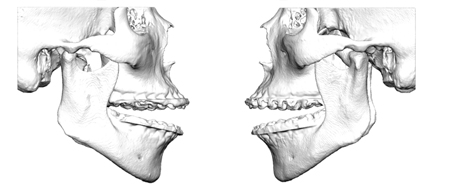
Fig. 15

Fig. 16

One week prior to surgery the patient underwent CT angiography and endovascular arterial embolization of the left internal maxillary arterial system. (Figs 8,9)
Fig. 8
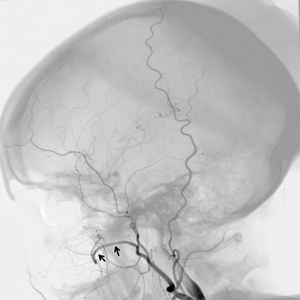
Fig. 9

Surgical Procedure (Figs 10-13, 17, 18, 20 & 21)
The surgery was carried out under nasoendotracheal intubation and general anesthesia. The blood pressure was intentionally lowered to minimize bleeding. The left TMJ and mandible was exposed via a hemi-coronal incision combined with a retro-submandibular incision. The facial nerve was exposed to localize and protect it. Continuous Facial Nerve monitoring with intermittent stimulation was used to identify the various branches. The zygomatic arch was resected. The condyle and tumor were removed. No untoward bleeding occurred. The wound was temporarily closed. Attention was directed intra-orally and a right sagittal split osteotomy was performed. The occlusion was corrected with the aid of a custom splint. The right ramus was then stabilized with 3 osteosynthesis screws. The intraoral wound was closed.
Fig. 10
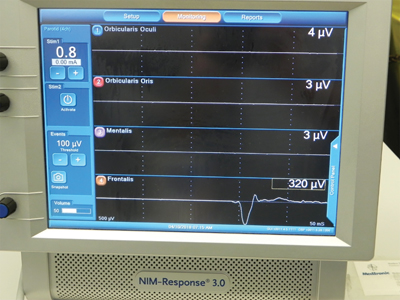
Fig. 11

Fig. 12
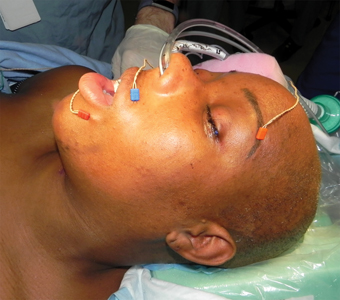
Fig. 13
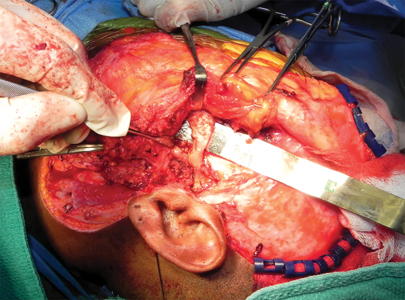
Attention was returned to the left side. The custom hybrid glenoid fossa-zygoma prosthesis and condylar prosthesis were installed with accurate fit. The wounds were closed. Bleeding was minimal. No severe bleeding occurred. Transfusion was not necessary.
The mandible was manipulated while still under anesthesia. There was good range of motion and stable occlusal alignment. The patient was taken to the intensive care unit where she was kept intubated and ventilated for the first postoperative night. This was done to protect the airway in case of severe swelling. Post- operative care included intravenous antibiotics and steroids.
The patient was extubated the first postoperative day. She had initial weakness of the left frontal branch of the facial nerve. This was managed with tear gel and eyelid taping. She also had partial numbness of the right lower lip and chin. The Facial Nerve and Inferior Alveolar Nerve were both seen to be visually intact at surgery. It was therefore thought this was a neuropraxia which would resolve on its own with time. An oculoplastic surgeon was consulted. He agreed with waiting and watching while keeping the eye lubricated. (indeed this proved to be the case with near return of normal function of the facial nerve and normal feeling to the right lower lip after 6 months).
Fig. 17

Fig. 18
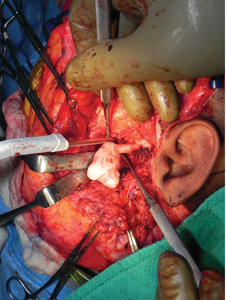
Fig. 20

Fig. 21

The patient was discharged on the fifth postoperative day in stable condition.
Postoperative histology confirmed the diagnosis of osteochondroma of the left mandibular condyle.
She was followed in the primary surgeon’s office on a regular basis. Bilateral orthodontic elastics were used for 3 months. Range of motion exercises were prescribed. The patient was prescribed a Therabyte jaw exerciser. Within 3 months she was completely pain free.
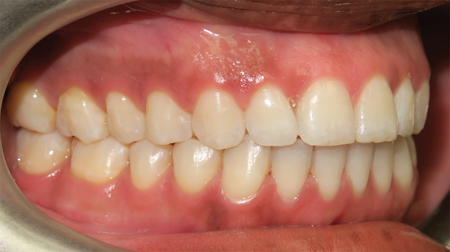
The most recent follow up was at 18 months post operatively. She has a stable occlusion with a pain free interincisal opening of 45 mm. (Figs 22-29)
Fig. 22

Fig. 23

Fig. 24

Fig. 25

Fig. 26

Fig. 27
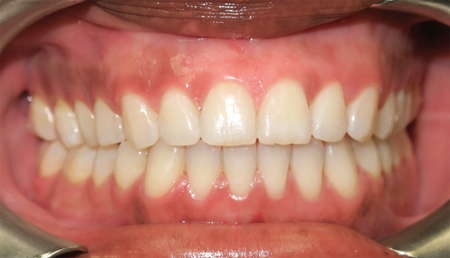
Fig. 28

Fig. 29
Discussion and Literature Review
Although rare, altered growth patterns of the mandible may occur in all of its regions including the condyle, ramus, and coronoid process. Unilateral growth of the mandible can result in significant asymmetry, malocclusion and may or may not be accompanied by pain.1 Osteochondromas represent 35% to 50% of all bone tumors, making it the most common benign tumor of bone. Osteochondromas in the oral and maxillofacial region comprises approximately 1% of all osteochondromas.2 This benign tumor may occur on any bone that forms via endochondral ossification and can be found in all bones of the maxillofacial skeleton.3,4 Within the craniofacial complex, the most frequent location of osteochondroma growth is located on the coronoid process or the condyle.4
The clinical presentation of patients with underlying osteochondromas can vary from patient to patient. Clinical signs may include progressive mandibular asymmetry, trismus, dysphonia, as well as the presence of a malocclusion.5 The diagnosis of osteochondromas of the craniofacial complex is best completed through clinical examination, radiographic imaging and correlation to histological features.6 Radiographic imaging may include plain films (panoramic), bone scintigraphy, computed tomography (CT), as well as magnetic- resonance imaging (MRI).7 Osteochondromas on panoramic examinations appear radiopaque, with the cortex and medullary cavity merging with the underlying bone.8
The pathophysiology of osteochondromas remains controversial in the literature as the debate exists whether these lesions are true tumors or alterations in development.9 Historically, the belief was that osteochondromas formed from the remaining components of the cartilaginous primordial cranium.10 It was proposed that the tumor resulted from defects in the periosteal cuff and herniation of the epiphyseal plate cartilage during TMJ development.2 In 1999 however, Porter et al., described genetic basis of the neoplastic pathogenesis in osteochondromas.11 Identifications of deletions of q24 from chromosomes 8 and p 11-13 from chromosome 11 from cytogenic analysis have been found to be present in patients with solitary and multiple hereditary osteochondromas.4 These loci have been described as EXT1 (8q24) and EXT2 (11P11) respectively. The solitary type is believed to develop after two successive mutations to the EXT gene stimulating neoplastic growth. Although rare, it is believed that the progression of osteochondromas to cancerous chondrosarcomas may be the result of additional mutations to these genes. The causation behind the genetic changes is believed to be radiation induced.5
Histologically, an osteochondroma will consist of trabecular bone with a rim of cartilage.8 Chondrocytes should appear in an organized fashion within columns of cartilage which are converting to bone.8 Outside of the cartilage columns, chondrocytes may be arranged in a less organized fashion.8 Adipose tissue and normal hematopoietic elements may be present between the bony trabeculae. The chondrocytes typically exhibit normal characteristics, although nuclear atypia may be observed.8 Solitary osteochondromas, are cited to have an approximately a 1% risk of malignant transformation.2
Treatment options for a condylar osteochondroma include both conservative and surgical considerations. The decision as to which approach to take is heavily reliant on the dynamic growth of the osteochondroma. Some osteochondromas have shown that they may stabilize with time, and in these cases conservative therapy may include non-treatment or surgery to correct cosmetic and masticatory dysfunction.1 In these cases, interval growth can be assessed with serial imaging for growth changes, or through scintigraphy to identify active growth of the bone in the region.12 If active growth is identified clinically (progressive open bite) and/or radiographically, surgical therapy maybe recommended for more favorable outcomes. Surgical considerations would include the necessity for ablation of the existing tumor, as well as correction of the malocclusion. This treatment often will include a combination of temporomandibular joint surgery and orthognathic surgery.13
In the case of surgical management of osteochondromas, tumor principles require removal of the neoplasm proper as well as a bone margin to minimize tumor recurrence. The spectrum of surgical treatment as such in literature includes subtotal condylectomy as well as total condylectomy for tumor ablation.13 As the primary goal of surgery is to excise the lesion in total, Wolford has described that incomplete removal during the condylectomy procedure may result in tumor recurrence.14 Peroz and colleagues have extensively reviewed the literature on comparing conservative osteochondroma excision only vs. condylectomy for tumor removal. In a review of 34 cases, 26/26 cases treated with condylectomy remained recurrence free while 2/9 cases treated with tumor excision only demonstrated recurrence.15 Total condylectomy as such would provide improved surgical margins, and Kim et al., demonstrated in their 18 year follow up study of 27 joints, the absence of any recurrences when osteochondromas were treated with condylectomy.16 Patients in this study were followed with bone scans and clinical evaluation. These findings are consistent with other published literature with recurrence rates reported to be less than 2% post condylectomy.4,17
Most often management of osteochondromas includes condylectomy as well as immediate reconstruction. Conservative reconstruction has been described in literature to include lesion excision with mandibular condyle reshaping 4, 17 and high condylectomy with condylar head reshaping.14 In these cases, concurrent disc plication was often performed. The advantage of these procedures is the maintenance of the native mandible and bone contact with the articular disc.4,14,17 Long term outcomes in these cases however may still need to be evaluated given the reported risk of recurrence with conservative treatment and the described risk of malignant transformation of osteochondromas.15,18
Reconstruction options for cases treated with total condylectomy include ramus osteotomies and reshaping,19 replacement with non-vascularized grafts (costochondral, iliac crest, metatarsal, coronoid process), vascularized grafts (fibula), as well as alloplastic replacement total joint replacement.20 The costochondral graft was first described in 1920 by Gillies and has been a staple of temporomandibular joint replacement in facial reconstruction. This grafting procedure has traditionally been favorable due to its low morbidity, biological compatibility and more importantly the ability of the graft to undergo functional adaption. The functional adaptability of costochondral grafts is one of the primary reasons this graft is utilized in the pediatric population to
facilitate continued growth.21
Temporomandibular joint reconstruction with the use of alloplastic materials provides the ability to reconstruct large, complex defects without the need to harvest tissue from a distant donor site. The alloplastic total joint replacements allow the transfer of functional forces to supporting bone structures with the minimization of any micromotion.22 Alloplastic joint replacement technology has been an ever-evolving entity, and custom (patient fitted) joint replacements can now be designed and fabricated. The prosthetic design process allows for the incorporation of occlusal corrections, and adaptation to anatomical deformities. It has been utilized in the treatment of numerous clinical pathologies of the TMJ including degenerative joint disease, ankylosis, chronic pain, and agenesis in addition to reconstruction for tumor management. The author of this article recently has published 5-year data on alloplastic TJR reviewing over 62 joints replaced with the Biomet Micro fixation system. This review demonstrated a 91% satisfaction rate with significant improvements in maximal incisal opening and masticatory efficiency.23 Other studies with larger numbers and longer follow up are ongoing, Long term data of total joint replacements has also been encouraging, showing minimal wear and failure of the prosthetics.24,25
Summary
This paper presents a case of a patient with a large osteochondroma of the mandibular condyle. The patient had a successful outcome with a collaborative multidisciplinary approach. This included orthodontics, interventional neuroradiology, otolaryngology-head and neck oncology surgery and oral and maxillofacial surgery. A patient fitted custom TMJ prosthesis was made using digital work flow design and CAD-CAM technolgy. There is ever increasing evidence to support the use of alloplastic devices for total TMJ replacemeent as the procedure of choice in adult patients.
Oral Health welcomes this original article.
Disclaimer: The Authors have no conflict of interest to declare in the submission of this paper.
References
- Holmlund, A. B.; Gynther, G. W.; Reinholt, F. P., Surgical treatment of osteochondroma of the mandibular condyle in the adult: A 5-year follow-up. International Journal of Oral and Maxillofacial Surgery 2004, 33 (6), 549-553.
- Saito, T.; Utsunomiya, T.; Furutani, M.; Yamamoto, H., Osteochondroma of the mandibular condyle : a case report and review of the literature. Journal of Oral Science 2001, 43 (4), 293-297.
- Dominguez, M. F.; Castillo, J. L. D.; Guerra, M. M.; Sanchez, R. S.; De La Plata, M. M., Condylar Osteochondroma Treated with Total Condylectomy and Preservation of the Articular Disc: A Case Report. Craniomaxillofac Trauma Reconstruction 2015, 8, 136-140.
- Aydιn, M. A.; Küçükçelebi, A.; Sayιlkan, S.; Çelebioğlu, S., Osteochondroma of the mandibular condyle: Report of 2 cases treated with conservative surgery. Journal of Oral and Maxillofacial Surgery 2001, 59 (9), 1082-1089.
- Harper, G. D.; Dicks-Mireaux, C.; Leiper, A. D., Total body irradiation induced osteochondromata. Journal of Pediatric Orthopaedics 1998, 18 (3), 356-358.
- Dandriyal, R.; Giri, K. Y.; Pant, S.; Alam, S.; Joshi, A., Giant Osteochondroma of the Coronoid Process. Journal of Oral and Maxillofacial Surgery 2015, 14, 412-416.
- Sekhar, M. M.; Loganathan, S., Giant osteochondroma of the mandibular condyle. Journal of oral and Maxillofacial Pathology 2015, 19 (3), 407.
- B.F., W.; Luna, M. A.; Newland, R. J., Temporomandibular Joint Neoplasms and Pseudotumors. Advances in Anatomic Pathophysiology 2000, 7 (6), 365-381.
- Devaney, K., Dahlin’s Bone Tumors. General Aspects and Data on 11,087 Cases The American Journal of Surgical Pathology 1996, 20 (10), 1298.
- Virchow, R., Ueber multiple Exostosen,mit Vorlegung von Praparaten. Berl.Klin.Wochenschr. 1891, 28, 1082.
- Porter, D. E.; Simpson, A. H. R. W., The neoplastic pathogenesis of solitary and multiple osteochondromas. The Journal of Pathology 1999, 188 (2), 119-125.
- Hodder, S. C.; Rees, J. I. S.; Oliver, T. B.; Facey, P. E.; Sugar, A. W., SPECT bone scintigraphy in the diagnosis and management of mandibular condylar hyperplasia. British Journal of Oral and Maxillofacial Surgery 2000, 38 (2), 87-93.
- Wolford, L. M.; Mehra, P.; Franco, P., Use of conservative condylectomy for treatment of osteochondroma of the mandibular condyle. Journal of Oral and Maxillofacial Surgery 2002, 60 (3), 262-268.
- Wolford, L. M.; Movahed, R.; Dhameja, A.; Allen, W. R., Low Condylectomy and Orthognathic Surgery to Treat Mandibular Condylar Osteochondroma: A Retrospective Review of 37 Cases. Journal of Oral and Maxillofacial Surgery 2014, 72 (9), 1704-1728.
- Peroz, I.; Scholman, H. J.; Hell, B., Osteochondroma of the mandibular condyle: a case report. International Journal of Oral and Maxillofacial Surgery 2002, 31 (4), 455-456.
- Kim, J. Y.; Ha, T. W.; Park, J. H.; Jung, H. D.; Jung, Y. S., Condylectomy as the treatment for active unilateral condylar hyperplasia of the mandible and severe facial asymmetry: retrospective review over 18 years. International Journal of Oral and Maxillofacial Surgery 2019, 48 (12), 1542-1551.
- Ortakoglu, K.; Akcam, T.; Sencimen, M.; Karakoc, O.; Ozyigit, H. A.; Bengi, O., Osteochondroma of the mandible causing severe facial asymmetry: A case report. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 2007, 103 (5), e21-e28.
- Barrett, A. W.; Hopper, C.; Speight, P. M., Oral presentation of secondary chondrosarcoma arising in osteochondroma of the nasal septum. International Journal of Oral and Maxillofacial Surgery 1996, 25 (2), 119-121.
- Loftus, M. J.; Bennett, J. A.; Fantasia, J. E., Osteochondroma of the mandibular condyles. Oral Surgery, Oral Medicine, Oral Pathology 1986, 61 (3), 221-226.
- Vasconcelos, B. C.; Porto, G. G.; Bessa-Nogueria, R. V.; Nascimento, M. M., Surgical treatment of temporomandibular joint ankylosis: follow-up of 15 cases and literature review. Med Oral Patol Oral Cir Bucal 2009, 14 (1), E34-E38.
- Kaban, L. B.; Perrott, D. H.; Fisher, K., A protocol for management of temporomandibular joint ankylosis. Journal of Oral and Maxillofacial Surgery 1990, 48 (11), 1145-1151.
- Mercuri, L. G., Condyle replacement after tumor resection: comparison of individual prefabricated titanium implants and costochondral grafts. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology and Endodontology, 2009, 108 (2), 153-155.
- Boyo, A.; McKay, J.; Lebovic, G.; Psutka, D. J., Temporomandibular joint total replacement using the Zimmer Biomet Microfixation patient-matched prosthesis results in reduced pain and improved function. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology 2019, 128 (6), 572-580.
- Quinn, P: Twenty Year Prospective Study on the Biomet Microfixation Stock TMJ Total Joint Prosthesis. Data published in a webinar, 2015
Wolford,L; Mercuri,L; Schneiderman, E; Movahed,R; Allen,W; Twenty Year Follow Up Study on a Patient Fitted Temporomandibular Joint Prosthesis; The Techmedica/TMJ Concepts - Device. Journal of Oral and Maxillofacial Surgery, May, 2015. 73:952-960.
About the Authors
Jakob Nickerson, Hospital Dental Resident, Sunnybrook Hospital, Toronto. Peter Ta, Attending Oral and Maxillofacial Surgeon, Sunnybrook Hospital and Trillium Hospital, Toronto, Private Practice, Fountain View Oral, Facial and Implant Surgery, Mississauga, ON.
Thomas Kertesz, BSc, MSc, DDS, FRCD(C), Attending Oral and Maxillofacial Surgeon, Trillium Hospital, Toronto, Private practice, Fountain View Oral, Facial and Implant Surgery, Mississauga, ON.
Fred Murrell, Private practice, Village Orthodontics, Toronto, ON, Diplomat, American Board of Orthodontics. Walter Montanera MD FRCP(C), Diagnostic and Therapeutic Neuroradiology, Associate Professor, Faculty of Medicine, University of Toronto, Dept. of Medical Imaging St. Michael’s Hospital; University of Toronto, ON.
Ian J. Witterick, Professor and Chair Department of Otolaryngology-Head & Neck Surgery, Faculty of Medicine, University of Toronto, Otolaryngologist-in-Chief, Sinai Health System, Toronto, ON.
Marco Caminiti, Assistant Professor and Program Head, Oral and Maxillofacial Surgery, Faculty of Dentistry, University of Toronto. Co-director, University of Toronto Fellowship Program in Advanced TMJ and Orthognathic Surgery, Head, Division of Oral and Maxillofacial Surgery, Humber Regional Hospital, Toronto Head, Division of Oral and Maxillofacial Surgery, Bloorview Hospital for Children, Toronto.
David J. Psutka, Assistant Professor, Oral and Maxillofacial Surgery, Faculty of Dentistry, University of Toronto, Senior Surgeon, Mount Sinai Hospital Centre of Excellence for Advanced TMJ Reconstructive Surgery, o-director, University of Toronto Fellowship Program in Advanced TMJ and Orthognathic Surgery, Private Practice, Fountain View Oral, Facial and Implant Surgery, Mississauga.
To view more articles from the 2020 Oral Surgery issue, please click here.













